Genetic Variation Associated with Influenza Severity Identified
|
By LabMedica International staff writers Posted on 03 Aug 2017 |
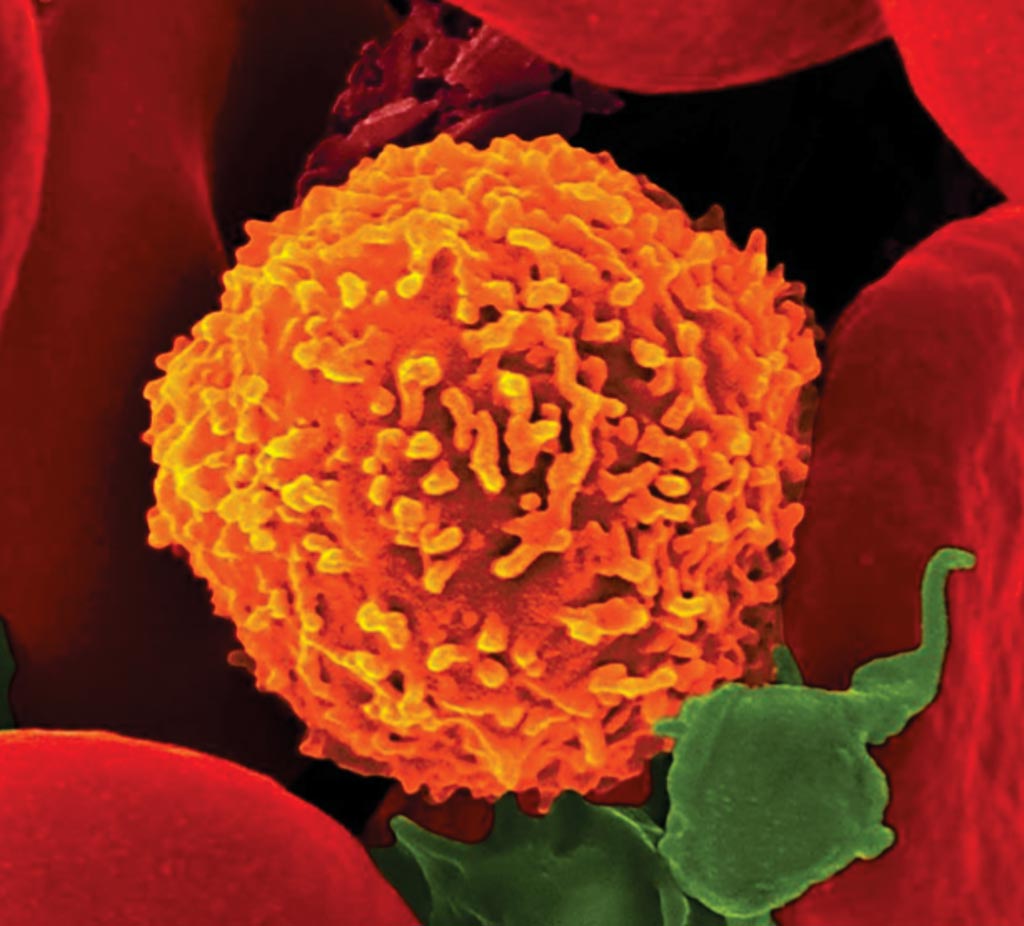
Image: A scanning electron micrograph of a CD8+ T cell engaging a virus (Photo courtesy of Dennis Kunkel).
It is estimated that in the USA influenza -related deaths in recent years have ranged from 12,000 to 56,000. Factors like age, obesity, pregnancy and such chronic health conditions as asthma, chronic lung disease and heart disease are associated with an elevated risk of complications and death.
However, there are no proven genetic markers of influenza risk with an established mechanism of action. Interferon Induced Transmembrane Protein 3 (IFITM3) is an anti-viral protein that helps to block influenza infection of lung cells and to promote survival of the killer T cells that help clear the infection in the airways.
A group of scientists collaborating with those at St. Jude Children's Research Hospital (Memphis, TN, USA) searched for other possible IFITM3 variants that correlated with gene expression, levels of the IFITM3 proteins and were common in influenza patients in the USA. The search led to an IFITM3 variant known as rs34481144. They checked 86 children and adults in Memphis with confirmed influenza infections and found two-thirds of patients with the most severe symptoms carried at least one copy of the newly identified high-risk IFITM3 variant. The high-risk variant was found in just 32% of patients with milder symptoms.
The team also found an association between the newly identified high-risk variant and severe and fatal influenza infections in 265 critically ill pediatric patients hospitalized in one of 31 intensive care units nationwide. The patients did not have health problems that put them at high risk for severe influenza. Of the 17 patients in this group who died from the infection, 14 carried at least one copy of the newly identified high-risk variant. Further study revealed how binding differed between the high-risk and protective variants. Those differences led to lower levels of the IFITM3 protein in individuals with two copies of the high-risk gene variant compared to other patients. The Memphis influenza patients also had fewer of the killer T cells in their upper airways. The study identifies a new regulator of IFITM3 expression that associates with CD8+ T cell levels in the airways and a spectrum of clinical outcomes.
Paul Thomas, PhD, an immunologist and corresponding author of the study, said, “A genetic marker of influenza risk could make a life-saving difference, particularly during severe influenza outbreaks, by helping prioritize high-risk patients for vaccination, drug therapy and other interventions. These results raise hopes that this newly identified IFITM3 variant might provide such a marker.” The study was published on July 17, 2017, in the journal Nature Medicine.
Related Links:
St. Jude Children's Research Hospital
However, there are no proven genetic markers of influenza risk with an established mechanism of action. Interferon Induced Transmembrane Protein 3 (IFITM3) is an anti-viral protein that helps to block influenza infection of lung cells and to promote survival of the killer T cells that help clear the infection in the airways.
A group of scientists collaborating with those at St. Jude Children's Research Hospital (Memphis, TN, USA) searched for other possible IFITM3 variants that correlated with gene expression, levels of the IFITM3 proteins and were common in influenza patients in the USA. The search led to an IFITM3 variant known as rs34481144. They checked 86 children and adults in Memphis with confirmed influenza infections and found two-thirds of patients with the most severe symptoms carried at least one copy of the newly identified high-risk IFITM3 variant. The high-risk variant was found in just 32% of patients with milder symptoms.
The team also found an association between the newly identified high-risk variant and severe and fatal influenza infections in 265 critically ill pediatric patients hospitalized in one of 31 intensive care units nationwide. The patients did not have health problems that put them at high risk for severe influenza. Of the 17 patients in this group who died from the infection, 14 carried at least one copy of the newly identified high-risk variant. Further study revealed how binding differed between the high-risk and protective variants. Those differences led to lower levels of the IFITM3 protein in individuals with two copies of the high-risk gene variant compared to other patients. The Memphis influenza patients also had fewer of the killer T cells in their upper airways. The study identifies a new regulator of IFITM3 expression that associates with CD8+ T cell levels in the airways and a spectrum of clinical outcomes.
Paul Thomas, PhD, an immunologist and corresponding author of the study, said, “A genetic marker of influenza risk could make a life-saving difference, particularly during severe influenza outbreaks, by helping prioritize high-risk patients for vaccination, drug therapy and other interventions. These results raise hopes that this newly identified IFITM3 variant might provide such a marker.” The study was published on July 17, 2017, in the journal Nature Medicine.
Related Links:
St. Jude Children's Research Hospital
Latest Immunology News
- AI Predicts Tumor-Killing Cells with High Accuracy
- Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
- AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
- Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
- Testing Method Could Help More Patients Receive Right Cancer Treatment
- Groundbreaking Test Monitors Radiation Therapy Toxicity in Cancer Patients
- State-Of-The Art Techniques to Investigate Immune Response in Deadly Strep A Infections
- Novel Immunoassays Enable Early Diagnosis of Antiphospholipid Syndrome
- New Test Could Predict Immunotherapy Success for Broader Range Of Cancers
- Simple Blood Protein Tests Predict CAR T Outcomes for Lymphoma Patients
- Cell Sorter Chip Technology to Pave Way for Immune Profiling at POC
- Chip Monitors Cancer Cells in Blood Samples to Assess Treatment Effectiveness
- Automated Immunohematology Approaches Can Resolve Transplant Incompatibility
- AI Leverages Tumor Genetics to Predict Patient Response to Chemotherapy
- World’s First Portable, Non-Invasive WBC Monitoring Device to Eliminate Need for Blood Draw
- Predictive T-Cell Test Detects Immune Response to Viruses Even Before Antibodies Form
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more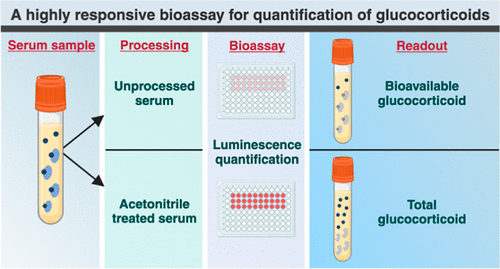
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel.jpg)
AI Predicts Tumor-Killing Cells with High Accuracy
Cellular immunotherapy involves extracting immune cells from a patient's tumor, potentially enhancing their cancer-fighting capabilities through engineering, and then expanding and reintroducing them into the body.... Read more
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read moreMicrobiology
view channel
Integrated Solution Ushers New Era of Automated Tuberculosis Testing
Tuberculosis (TB) is responsible for 1.3 million deaths every year, positioning it as one of the top killers globally due to a single infectious agent. In 2022, around 10.6 million people were diagnosed... Read more
Automated Sepsis Test System Enables Rapid Diagnosis for Patients with Severe Bloodstream Infections
Sepsis affects up to 50 million people globally each year, with bacteraemia, formerly known as blood poisoning, being a major cause. In the United States alone, approximately two million individuals are... Read moreEnhanced Rapid Syndromic Molecular Diagnostic Solution Detects Broad Range of Infectious Diseases
GenMark Diagnostics (Carlsbad, CA, USA), a member of the Roche Group (Basel, Switzerland), has rebranded its ePlex® system as the cobas eplex system. This rebranding under the globally renowned cobas name... Read more
Clinical Decision Support Software a Game-Changer in Antimicrobial Resistance Battle
Antimicrobial resistance (AMR) is a serious global public health concern that claims millions of lives every year. It primarily results from the inappropriate and excessive use of antibiotics, which reduces... Read morePathology
view channel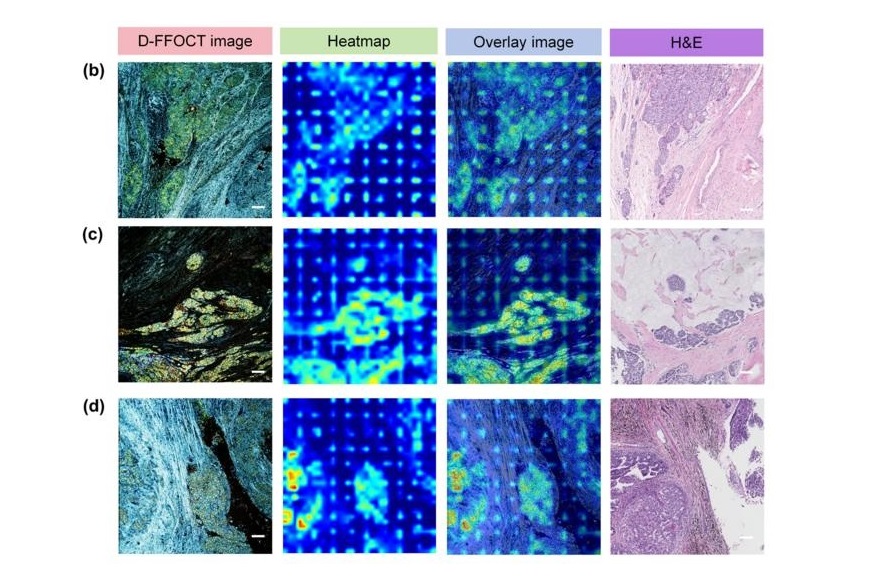
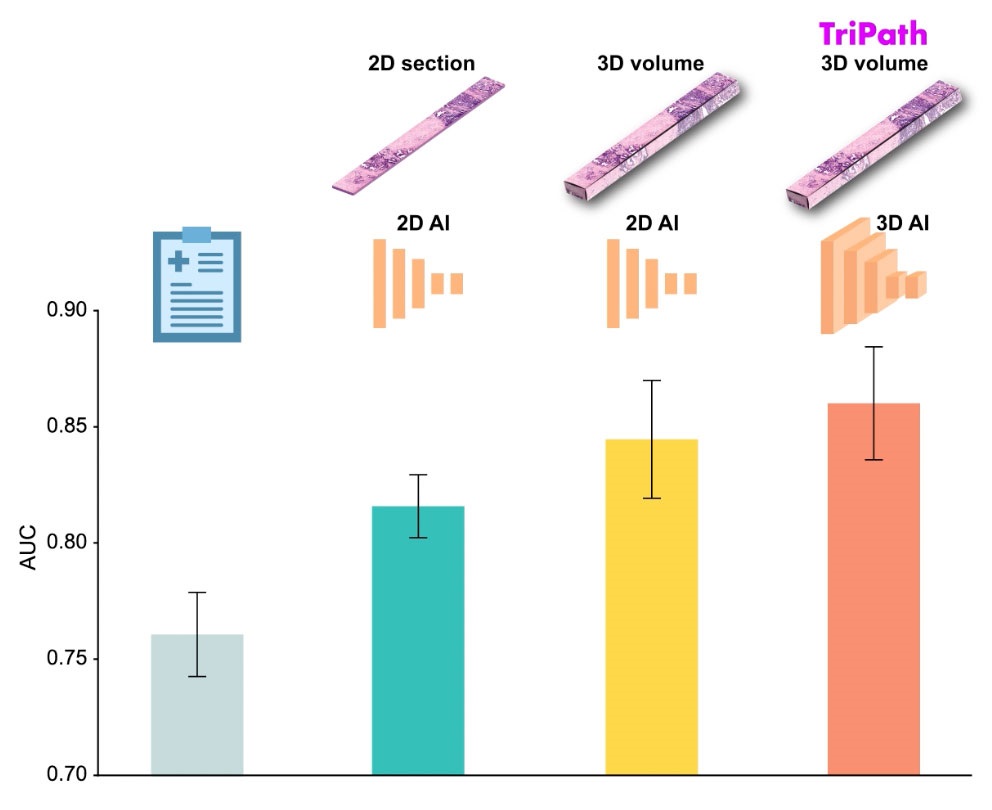
AI Integrated With Optical Imaging Technology Enables Rapid Intraoperative Diagnosis
Rapid and accurate intraoperative diagnosis is essential for tumor surgery as it guides surgical decisions with precision. Traditional intraoperative assessments, such as frozen sections based on H&E... Read more
HPV Self-Collection Solution Improves Access to Cervical Cancer Testing
Annually, over 604,000 women across the world are diagnosed with cervical cancer, and about 342,000 die from this disease, which is preventable and primarily caused by the Human Papillomavirus (HPV).... Read moreHyperspectral Dark-Field Microscopy Enables Rapid and Accurate Identification of Cancerous Tissues
Breast cancer remains a major cause of cancer-related mortality among women. Breast-conserving surgery (BCS), also known as lumpectomy, is the removal of the cancerous lump and a small margin of surrounding tissue.... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more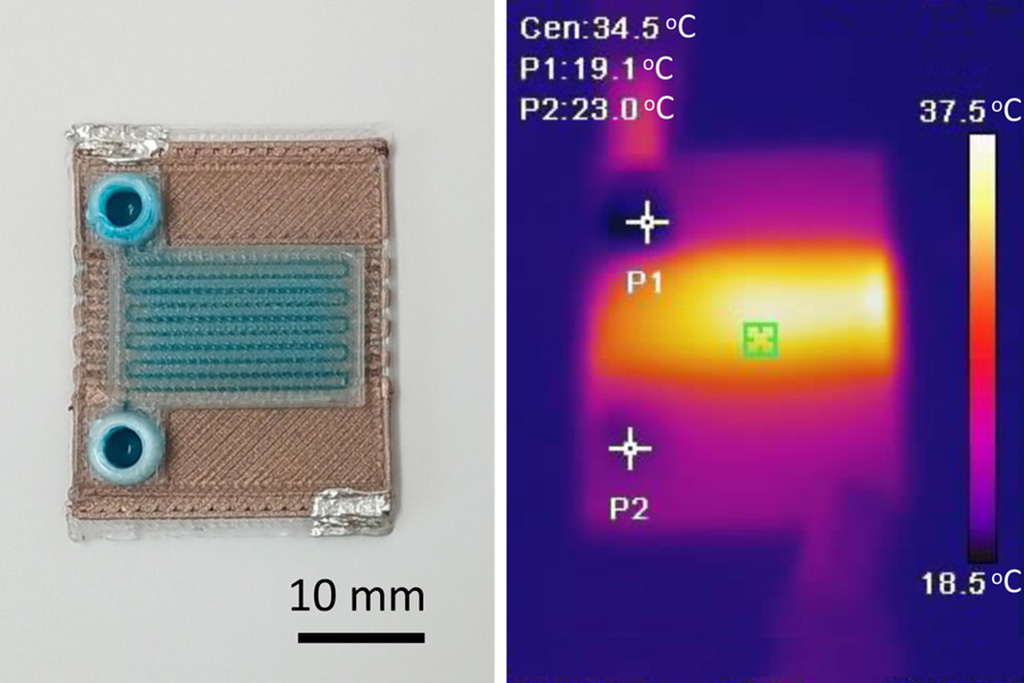
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel
Danaher and Johns Hopkins University Collaborate to Improve Neurological Diagnosis
Unlike severe traumatic brain injury (TBI), mild TBI often does not show clear correlations with abnormalities detected through head computed tomography (CT) scans. Consequently, there is a pressing need... Read more
Beckman Coulter and MeMed Expand Host Immune Response Diagnostics Partnership
Beckman Coulter Diagnostics (Brea, CA, USA) and MeMed BV (Haifa, Israel) have expanded their host immune response diagnostics partnership. Beckman Coulter is now an authorized distributor of the MeMed... Read more_1.jpg)