Hologic Highlights Automated Lab Solutions and Expansive Diagnostic Menu at AACC 2022
|
By LabMedica International staff writers Posted on 26 Jul 2022 |

Hologic, Inc. (Marlborough, MA, USA) will be demonstrating how its automated solutions and expansive diagnostic menu can help laboratories grow at the 2022 AACC Clinical Lab Expo.
Hologic is highlighting its Panther Scalable Solutions that allow laboratories to expand their testing menu while adding on flexibility, capacity and walkaway time. The Panther system serves as the foundation of Hologic’s automation with the option to add on the Panther Fusion module, Panther Plus, Panther Link and Panther Trax. Panther Scalable Solutions provides laboratories an economical and scalable path to growth. Laboratories can select from a broad menu of Aptima and Panther Fusion assays that help guide patient care and enable early detection to run on scalable automation. With a menu and pipeline of world-class assays, laboratories can consolidate their molecular testing today and meet their growing needs tomorrow.
For instance, Hologic’s Panther Fusion SARS-CoV-2/Flu A/B/RSV assay and Novodiag RESP-4 molecular diagnostic test assays detect and differentiate four of the most prevalent respiratory viruses that can present with similar clinical symptoms: severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), influenza A (Flu A), influenza B (Flu B) and respiratory syncytial virus (RSV). The Panther Fusion SARS-CoV-2/Flu A/B/RSV assay is a high-performance cartridge-based assay that runs on the fully automated high-throughput Panther Fusion system. The Novodiag RESP-4 assay runs on the Novodiag system, the fully automated molecular diagnostic solution that delivers on-demand testing for infectious disease and antimicrobial resistance.
Also to be featured at the event is Hologic’s ThinPrep processors which are intuitive, versatile, and scalable solutions for laboratories of all volumes. The portfolio offers reliable, best-in-class systems that help cytology labs automate their processes – decreasing the need for hands-on labor and increasing laboratory efficiencies. And because every sample is a patient, Hologic’s automated chain-of-custody technology delivers confidence in results without sacrificing productivity. The company’s ThinPrep Pap test is the first FDA-approved, liquid-based cytology option in cervical disease screening and remains the preferred choice in Pap testing in the US.
Hologic is also highlighting its range of collection devices that support a comprehensive testing portfolio for women's health and infectious disease and have been rigorously developed for highly accurate and reliable results. These include diagnostic collection devices and transfer tubes, direct load capture cap collection kit, direct load tube collection kit and the Aptima multi-test swab.
Related Links:
Hologic, Inc.
Latest AACC 2022 News
- AACC 2022 Review: International Meetings Rebound After Suffering Long COVID Hiatus
- Faster Method Diagnoses Pediatric Urinary Tract Infections
- Tianlong Showcases Integrated PCR Lab Solutions at AACC 2022
- Cellavision Introduces New Workflow Solution for Low-Volume Hematology Labs at AACC 2022
- Advanced Instruments Introduces New Automated Osmometer at AACC 2022
- Innovative Smartphone and AI-Based Tests Featured at AACC 2022
- New Tests Presented at AACC 2022 to Solve Challenges in Children's Healthcare
- 10-Minute Sepsis Risk Stratification Test Introduced at AACC 2022
- New System Combining DNA/RNA Extraction and Smart PCR Setup Launched at AACC 2022
- Novel Small Volume Blood Collection Device Launched at AACC 2022
- AACC Disruptive Technology Award Finalists Tackle Cancer, Women's Health, and STDs
- Meridian Demonstrates State-of-the-Art Urea Breath Test System at AACC 2022
- Nikon Instruments Displays Latest Clinical Pathology Products at AACC 2022
- Seegene Showcases Latest Molecular Testing Solutions at AACC 2022
- Werfen Introduces New Acute Care Diagnostic Products for CVOR at AACC 2022
- PerkinElmer Demonstrates Vanadis NIPT Screening Platform Featuring Groundbreaking cfDNA Technology
Channels
Clinical Chemistry
view channel
Alzheimer’s Blood Biomarkers Linked to Early Cognitive Differences Before Dementia
Blood-based screening for Alzheimer’s disease offers a noninvasive, lower-cost alternative to brain imaging or spinal fluid testing, yet its ability to flag the earliest cognitive changes has been unclear.... Read more
Urine-Based Test Shows Promise for Autism Screening in Children
Autism spectrum disorder (ASD) is commonly diagnosed through behavioral assessments, which can involve long waits that delay intervention. Earlier identification is linked to better developmental outcomes,... Read moreMolecular Diagnostics
view channel
Blood-Based Assay Enables Noninvasive Monitoring of Sarcoma Immunotherapy Response
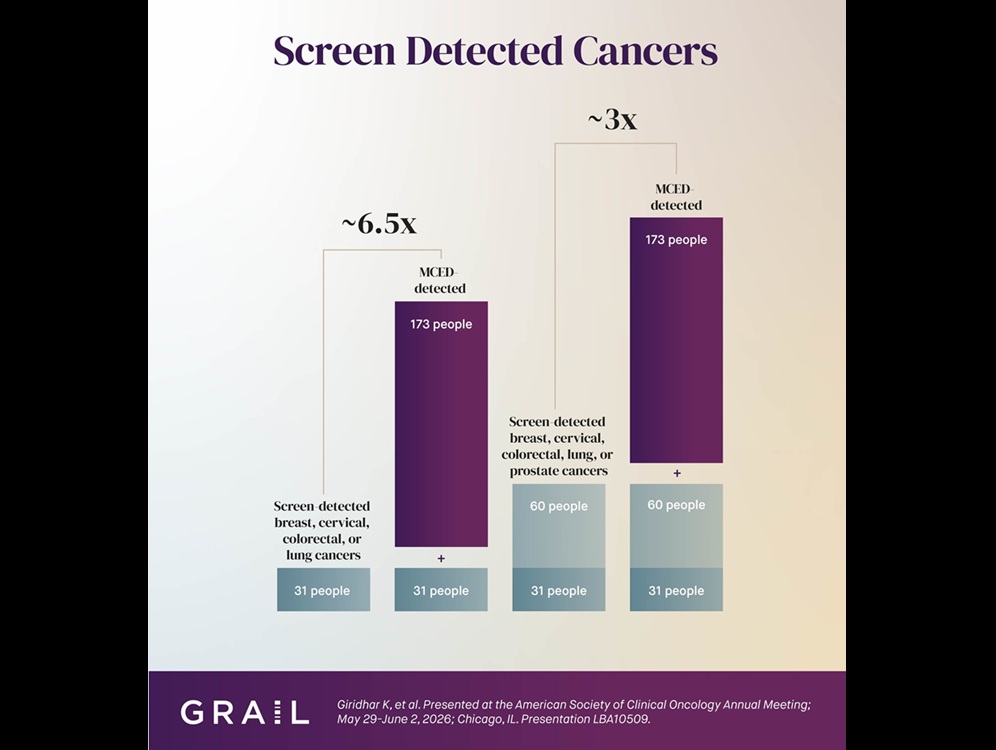
Sarcomas remain difficult to monitor during immunotherapy, as low tumor mutation burden can limit traditional circulating tumor DNA approaches and repeat tissue biopsies are often impractical in advanced disease.... Read more
Tumor Mutation Marker Helps Refine Lung Cancer Prognosis and Guide Therapy Selection
Lung cancer remains the leading cause of cancer death worldwide, while heterogeneous tumor genetics continue to complicate treatment decisions. Although molecular testing is increasingly used to match... Read moreHematology
view channel
Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
Sysmex America (Chicago, IL, USA) has introduced the next generation XR-Series, centered on the XR-10 Automated Hematology Module for high-complexity laboratories. The platform builds on the widely used... Read more
Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
Immune checkpoint inhibitors have improved outcomes across many cancers, yet only a subset of patients derive durable benefit and biomarkers to guide treatment remain limited. Eosinophils, best known for... Read moreImmunology
view channel
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
AI-Powered Atlas Maps Immune Structures Linked to Cancer Outcomes
Tertiary lymphoid structures are emerging as important indicators of antitumor immunity, but their heterogeneity and spatial context within tumors remain difficult to capture through routine diagnostics.... Read more
AI Tool Extracts Immune Signals from Biopsy to Inform Myeloma Therapy
Multiple myeloma is a bone marrow malignancy in which patients can respond very differently to the same treatments, making initial therapy decisions difficult. Clinicians must choose among options such... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel