Personalized CtDNA Analysis Detects Minimal Residual Disease
|
By LabMedica International staff writers Posted on 08 Jun 2021 |
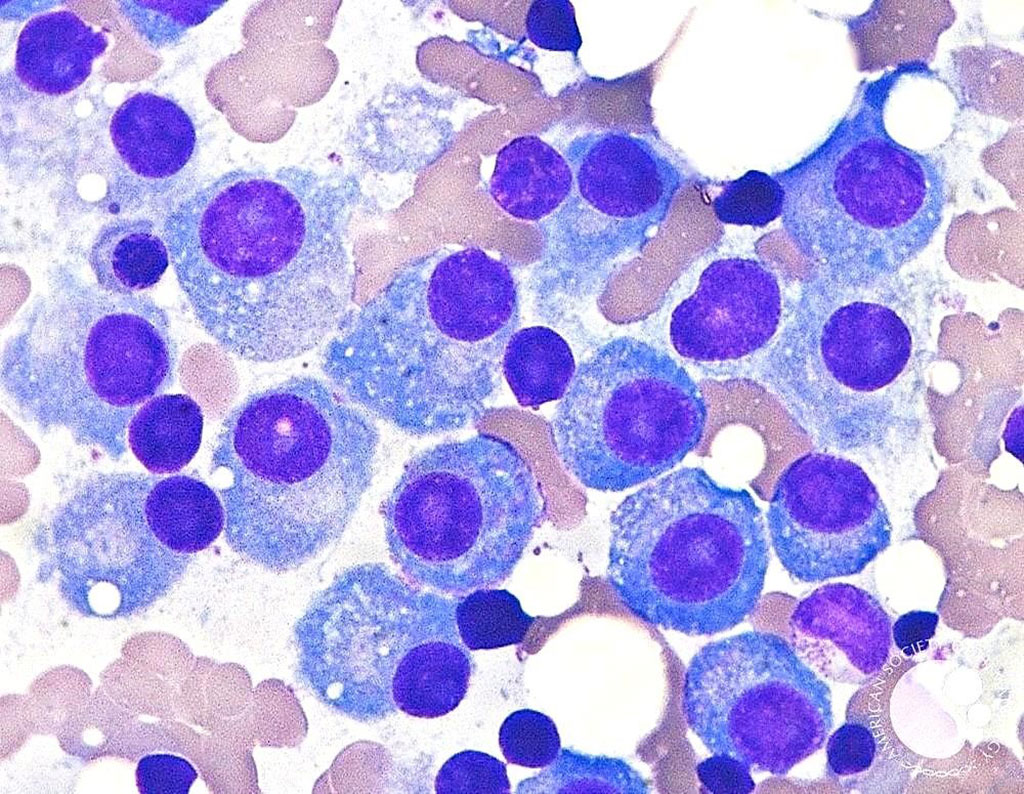
Image: Bone marrow aspirate showing mature plasma cells with eccentric nuclei and abundant basophilic cytoplasm indicative of multiple myeloma (Photo courtesy of Dr. David Israel Garrido, MD et.al.)
Multiple myeloma (MM), is a type of bone marrow cancer. It is called multiple myeloma as the cancer often affects several areas of the body, such as the spine, skull, pelvis and ribs. Minimal residual disease (MRD) is the name given to small numbers of leukemic cells that remain in the person during treatment, or after treatment when the patient is in remission.
Despite treatment with high-dose chemotherapy followed by autologous stem cell transplantation (AHCT), MM patients invariably relapse. MRD-negativity post-AHCT has emerged as the most important prognostic marker. Currently, MRD in MM is monitored via bone marrow aspirate sampling. Marrow MRD assays are limited by the spatial heterogeneity of marrow MM localization; extramedullary disease and sampling variability of marrow aspiration.
Hematologists at the Medical College of Wisconsin (Milwaukee, WI, USA) and their colleagues, analyzed in a retrospective, single-center study, circulating tumor DNA (ctDNA) MRD in blood samples collected from 28 patients with MM after upfront AHCT. A total of 80 plasma time points were available pre and post AHCT with a median follow-up of 92.4 months. Multiparameter flow cytometry (MFC) at 10-4 level was used to assess the MRD from the BM biopsy.
Individual bone marrow aspirates or Formalin-fixed, Paraffin-embedded (FFPE) slides from the time of MM diagnosis and matched normal blood were whole-exome sequenced, and somatic mutations were identified. MRD assessment at three months post-AHCT was performed by ctDNA analysis using a personalized, tumor-informed Signatera bespoke mPCR NGS assay (Natera Inc, San Carlos, CA, USA). The prognostic value of ctDNA was evaluated by correlating MRD status with clinical outcomes.
The scientists reported that ctDNA was detectable in 17/24 (70.8%) of pre-AHCT, 15/28 (53.6%) of ̃three months post-AHCT, and 11/28 (39.2%) of patients during the surveillance phase post-AHCT. Of the 15 ctDNA MRD positive patients, 93.3% experienced relapse on follow-up (hazard ratio: 5.64). Patients negative for ctDNA at three months post-AHCT had significantly superior progression-free survival (PFS) compared to positive (median PFS, 84 months versus 31 months) The positive predictive value (PPV) for relapse among patients positive for ctDNA at three months post-AHCT was 93.3%, and significantly higher than marrow multiparametric flow cytometry (MFC) of 68.4%.
The authors concluded that their study shows the feasibility that a tumor-informed assay on archival blood samples is predictive of relapse post-AHCT. Future prospective studies with real-time marrow next generation sequencing (NGS) and ctDNA samples are needed to define the role of ctDNA in MM and its prognostic significance. The study was presented at the virtual 2021 ASCO Annual Meeting held June 4-8, 2021.
Related Links:
Medical College of Wisconsin
Natera Inc
Despite treatment with high-dose chemotherapy followed by autologous stem cell transplantation (AHCT), MM patients invariably relapse. MRD-negativity post-AHCT has emerged as the most important prognostic marker. Currently, MRD in MM is monitored via bone marrow aspirate sampling. Marrow MRD assays are limited by the spatial heterogeneity of marrow MM localization; extramedullary disease and sampling variability of marrow aspiration.
Hematologists at the Medical College of Wisconsin (Milwaukee, WI, USA) and their colleagues, analyzed in a retrospective, single-center study, circulating tumor DNA (ctDNA) MRD in blood samples collected from 28 patients with MM after upfront AHCT. A total of 80 plasma time points were available pre and post AHCT with a median follow-up of 92.4 months. Multiparameter flow cytometry (MFC) at 10-4 level was used to assess the MRD from the BM biopsy.
Individual bone marrow aspirates or Formalin-fixed, Paraffin-embedded (FFPE) slides from the time of MM diagnosis and matched normal blood were whole-exome sequenced, and somatic mutations were identified. MRD assessment at three months post-AHCT was performed by ctDNA analysis using a personalized, tumor-informed Signatera bespoke mPCR NGS assay (Natera Inc, San Carlos, CA, USA). The prognostic value of ctDNA was evaluated by correlating MRD status with clinical outcomes.
The scientists reported that ctDNA was detectable in 17/24 (70.8%) of pre-AHCT, 15/28 (53.6%) of ̃three months post-AHCT, and 11/28 (39.2%) of patients during the surveillance phase post-AHCT. Of the 15 ctDNA MRD positive patients, 93.3% experienced relapse on follow-up (hazard ratio: 5.64). Patients negative for ctDNA at three months post-AHCT had significantly superior progression-free survival (PFS) compared to positive (median PFS, 84 months versus 31 months) The positive predictive value (PPV) for relapse among patients positive for ctDNA at three months post-AHCT was 93.3%, and significantly higher than marrow multiparametric flow cytometry (MFC) of 68.4%.
The authors concluded that their study shows the feasibility that a tumor-informed assay on archival blood samples is predictive of relapse post-AHCT. Future prospective studies with real-time marrow next generation sequencing (NGS) and ctDNA samples are needed to define the role of ctDNA in MM and its prognostic significance. The study was presented at the virtual 2021 ASCO Annual Meeting held June 4-8, 2021.
Related Links:
Medical College of Wisconsin
Natera Inc
Latest Hematology News
- Next-Generation Hematology Platform Streamlines High-Complexity Lab Workflows
- Blood Eosinophil Count May Predict Cancer Immunotherapy Response and Toxicity
- Higher Ferritin Threshold May Improve Iron Deficiency Detection in Children
- Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
- Advanced CBC-Derived Indices Integrated into Hematology Platforms
- Blood Test Enables Early Detection of Multiple Myeloma Relapse
- Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
- Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
- Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
- Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
- New Platelet Function Assay Enables Monitoring of Antiplatelet Therapy
- Open Multi-Omics Platform Identifies Prognostic Subtypes in Blood Cancers
- AI-Powered Digital Workflow Standardizes Bone Marrow Aspirate Morphology
- Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
- New Guidelines Aim to Improve AL Amyloidosis Diagnosis
- Automated Hemostasis System Helps Labs of All Sizes Optimize Workflow
Channels
Clinical Chemistry
view channel
Simple Dual-Tau Blood Test Detects and Stages Alzheimer’s Disease
Alzheimer’s disease is typically confirmed and staged with positron emission tomography scans and cerebrospinal fluid testing, procedures that are costly and invasive. Broader access to minimally invasive... Read more
Alzheimer’s Blood Biomarkers Linked to Early Cognitive Differences Before Dementia
Blood-based screening for Alzheimer’s disease offers a noninvasive, lower-cost alternative to brain imaging or spinal fluid testing, yet its ability to flag the earliest cognitive changes has been unclear.... Read moreMolecular Diagnostics
view channel
Genomic Test Guides Chemotherapy Decisions in Early-Stage Breast Cancer
Selecting adjuvant therapy for early-stage hormone receptor-positive breast cancer depends on accurately assessing long-term risk of distant recurrence. Clinical features alone can leave uncertainty about... Read more
Simple Cytogenetic Method Could Improve Classification of ALL Subtypes
Many cancers deviate from the normal chromosome number, but the clinical impact of extreme chromosome loss remains unclear. This widespread genomic disruption is associated with aggressive disease and... Read more
Blood-Based Assay Enables Noninvasive Monitoring of Sarcoma Immunotherapy Response
Sarcomas remain difficult to monitor during immunotherapy, as low tumor mutation burden can limit traditional circulating tumor DNA approaches and repeat tissue biopsies are often impractical in advanced disease.... Read moreImmunology
view channel
Study Points to Autoimmune Pathway Behind Long COVID Symptoms
Long COVID leaves many SARS-CoV-2 survivors with persistent fatigue, cognitive issues, palpitations, and musculoskeletal pain for months or years. Estimates cited in new research suggest 4%–20% of infected... Read more
Metabolic Biomarker Distinguishes Latent from Active Tuberculosis and Tracks Treatment Response
Tuberculosis (TB) remains the world’s leading infectious killer, with 10.8 million cases and 1.25 million deaths recorded globally in 2023. Yet many infected individuals never develop active disease, underscoring... Read moreMicrobiology
view channel
Gut Microbiome Signatures Help Identify Risk of IBD Progression
Inflammatory bowel disease (IBD), encompassing Crohn’s disease and ulcerative colitis, is a chronic relapsing inflammatory disorder of the gastrointestinal tract with highly variable outcomes.... Read more
FDA-Cleared Gastrointestinal Panel Detects 24 Pathogen Targets
Clinical guidelines support testing based on patient presentation in suspected gastrointestinal infections, yet available technologies have often forced laboratories to choose between panels that are too... Read morePathology
view channel
Blood-Based Method Tracks Gene Activity in the Living Brain
Real-time measurement of gene activity in the brain has been limited by assays requiring destructive tissue sampling. Tracking active genes could reveal how the body responds to environmental factors,... Read more
FDA Approval Expands Automated PD-L1 Testing Across Solid Tumors
Clinical laboratories play a central role in guiding immunotherapy by reporting programmed death ligand-1 (PD‑L1) status across multiple solid tumors. Many sites are standardizing this work on fully automated... Read moreTechnology
view channel
AI Platform Links Biomarker Results to Cancer Clinical Trials and Guidelines
Oncology teams must manage growing volumes of genomic data, rapidly evolving clinical trial options, and frequently updated care guidelines, all within tight clinic schedules. Translating complex tumor... Read more
Agentic AI Platform Supports Genomic Decision-Making in Oncology
Oncology care teams increasingly face the challenge of managing complex molecular diagnostics, evolving treatment options, and extensive electronic health record documentation. Translating multimodal data... Read moreIndustry
view channel