Novel Rapid Test Developed for Detecting Carbapenemase
|
By LabMedica International staff writers Posted on 02 Apr 2020 |

Image: The SpectraMax M5 Microplate Reader is the standard for UV/visible multi-mode reader absorbance, providing ultrafast, full spectral range detection for cuvettes, 96-, and 384-well microplates (Photo courtesy of Molecular Devices).
As a potent β-lactamase, carbapenemase can degrade almost all β-lactam antimicrobial drugs, including the carbapenems, regarded as the last line of therapy for many life-threatening infections. If uncontrolled, the spread of these carbapenemases is expected to increase therapeutic failure and leave many patients with no effective treatment options.
Despite the urgency, timely carbapenemase detection remains a challenge for microbiology laboratories. Phenotypic assays are inexpensive and easily performed, but their use requires 24–48 hours and many lack sensitivity or specificity. The widespread use of other assays (e.g., molecular tests of carbapenemase genes, mass spectrometry detection of carbapenem hydrolysis) is impeded by the expertise required to perform them and their cost.
Scientists affiliated with the Massachusetts General Hospital (Boston, MA, USA) demonstrate that by using fluorescence identification of β-lactamase activity (FIBA), carbapenemase production in bacteria can be detected sensitively and specifically in 10 minutes, with only one step. FIBA uses a dark fluorescence probe, β-LEAF (β-lactamase enzyme–activated fluorophore), which turns fluorescent when cleaved by β-lactamases, including penicillinases, extended-spectrum β-lactamases (ESBL), AmpC β-lactamases, and carbapenemases.
The team tested FIBA on 76 randomly selected infection isolates. To start the assay, 25 μL of 1 × 1010 CFU/mL bacterial suspension made by colonies grown overnight on BHI agar is added to each well. To monitor the increase rate, fluorescence measurement is then performed at 37 °C at 10-second intervals for 10 minutes with Ex/Em 450/510 nm in the Spectramax M5 plate reader (Molecular Devices, San Jose, CA, USA).
Genetic test results for β-lactam resistance were provided with the isolates. Among these, 55 were carbapenemase positive, carrying the major epidemic carbapenemase types including Klebsiella pneumoniae carbapenemase, imipenem-hydrolyzing β-lactamase, metallo-β-lactamase, New Delhi metallo-β-lactamase, oxacillinase, Serratia marcescens enzyme, São Paulo metallo-β-lactamase, Verona integron-encoded metallo-β-lactamase, and New Delhi metallo-β-lactamase oxacillinase. The other 21 isolates expressed noncarbapenemase β-lactamases.
The authors concluded that FIBA can be performed ≈10 times faster than the most rapid carbapenemase test commercially available while maintaining comparable sensitivity and specificity. Its automated analysis improves turnaround time and reduces operator variability. With a reagent cost/assay of approximately USD 1.00, FIBA is close in price to phenotypic tests but substantially faster and less labor intensive. The study was published in the April, 2020 issue of the journal Emerging Infectious Diseases.
Related Links:
Massachusetts General Hospital
Molecular Devices
Despite the urgency, timely carbapenemase detection remains a challenge for microbiology laboratories. Phenotypic assays are inexpensive and easily performed, but their use requires 24–48 hours and many lack sensitivity or specificity. The widespread use of other assays (e.g., molecular tests of carbapenemase genes, mass spectrometry detection of carbapenem hydrolysis) is impeded by the expertise required to perform them and their cost.
Scientists affiliated with the Massachusetts General Hospital (Boston, MA, USA) demonstrate that by using fluorescence identification of β-lactamase activity (FIBA), carbapenemase production in bacteria can be detected sensitively and specifically in 10 minutes, with only one step. FIBA uses a dark fluorescence probe, β-LEAF (β-lactamase enzyme–activated fluorophore), which turns fluorescent when cleaved by β-lactamases, including penicillinases, extended-spectrum β-lactamases (ESBL), AmpC β-lactamases, and carbapenemases.
The team tested FIBA on 76 randomly selected infection isolates. To start the assay, 25 μL of 1 × 1010 CFU/mL bacterial suspension made by colonies grown overnight on BHI agar is added to each well. To monitor the increase rate, fluorescence measurement is then performed at 37 °C at 10-second intervals for 10 minutes with Ex/Em 450/510 nm in the Spectramax M5 plate reader (Molecular Devices, San Jose, CA, USA).
Genetic test results for β-lactam resistance were provided with the isolates. Among these, 55 were carbapenemase positive, carrying the major epidemic carbapenemase types including Klebsiella pneumoniae carbapenemase, imipenem-hydrolyzing β-lactamase, metallo-β-lactamase, New Delhi metallo-β-lactamase, oxacillinase, Serratia marcescens enzyme, São Paulo metallo-β-lactamase, Verona integron-encoded metallo-β-lactamase, and New Delhi metallo-β-lactamase oxacillinase. The other 21 isolates expressed noncarbapenemase β-lactamases.
The authors concluded that FIBA can be performed ≈10 times faster than the most rapid carbapenemase test commercially available while maintaining comparable sensitivity and specificity. Its automated analysis improves turnaround time and reduces operator variability. With a reagent cost/assay of approximately USD 1.00, FIBA is close in price to phenotypic tests but substantially faster and less labor intensive. The study was published in the April, 2020 issue of the journal Emerging Infectious Diseases.
Related Links:
Massachusetts General Hospital
Molecular Devices
Latest Technology News
- AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
- Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
- Noninvasive Sputum Test Detects Early Lung Cancer
- New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
- Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
- Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
- Online Tool Supports Family Screening for Inherited Cancer Risk
- Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
- New Electronic Pipette Enhances Workflows with Touchscreen Control
- AI Model Outperforms Clinicians in Rare Disease Detection
- AI-Driven Diagnostic Demonstrates High Accuracy in Detecting Periprosthetic Joint Infection
- Blood Test “Clocks” Predict Start of Alzheimer’s Symptoms
- AI-Powered Biomarker Predicts Liver Cancer Risk
Channels
Clinical Chemistry
view channel
Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
Serum proteins underpin many routine tests used to detect inflammation, hormonal imbalance, cardiovascular disease, and metabolic disorders. Yet pediatric interpretation often relies on adult reference... Read more
Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
Alzheimer’s disease and related dementias develop over years, making it difficult to identify at-risk patients before symptoms appear. Clinicians therefore need widely available laboratory markers that... Read more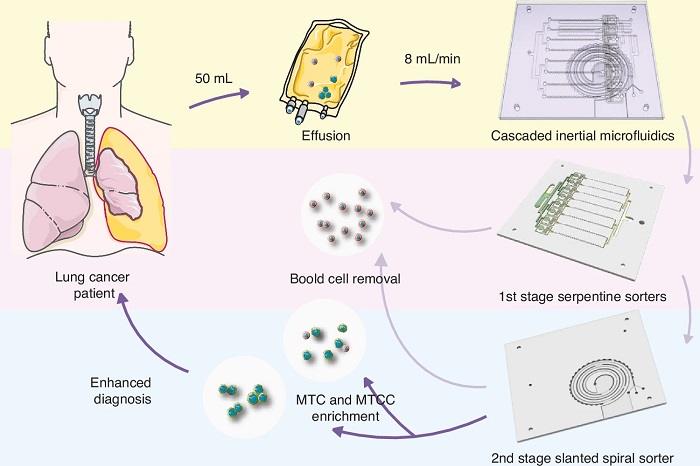
Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
Diagnosing malignancy from pleural effusion remains challenging because tumor cells are rare and clusters are easily disrupted during processing. Conventional cytology can miss malignant tumor cells and... Read moreMolecular Diagnostics
view channel
AI Blood Test Enhances Monitoring of Liver Cirrhosis Progression
Monitoring chronic liver disease remains difficult because clinicians rely on tools that can be inconsistent and may miss early progression. Standard approaches often combine ultrasound imaging with blood-based... Read more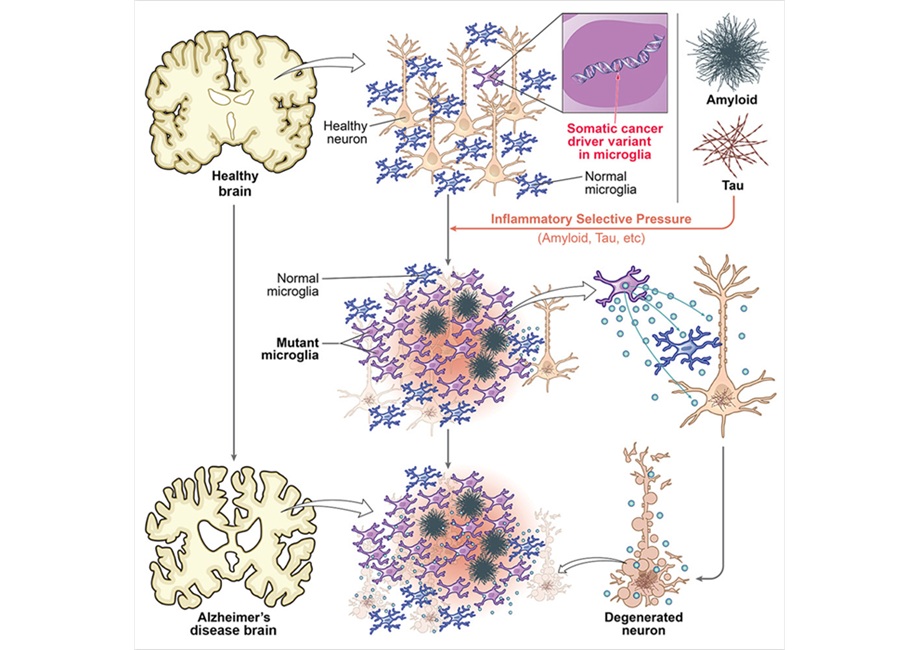
Cancer-Related Mutations in Immune Cells Linked to Alzheimer’s
Alzheimer’s disease is marked by protein aggregation and inflammatory changes in the brain’s immune system, yet its molecular drivers remain incompletely understood. With aging, human cells accumulate... Read more
Composite Blood Biomarkers Enable Early Detection of Common Cancers
Early diagnosis of colorectal, lung, and ovarian cancers remains challenging, with many patients identified only after tumors have begun to spread. A scalable blood test could expand access to screening,... Read more
Machine Learning Model Uses DNA Methylation to Predict Tumor Origin in Cancers of Unknown Primary
Cancers of unknown primary (CUP) are metastatic malignancies in which the primary site cannot be identified, complicating treatment selection. Many patients consequently receive broad, nonspecific chemotherapy... Read moreHematology
view channel
Single Assay Enables Rapid HLA and ABO Genotyping for Transplant Matching
CareDx (Brisbane, CA, USA) has introduced AlloSeq Nano, a nanopore‑based HLA (human leukocyte antigen) and ABO genotyping solution unveiled at the European Federation for Immunogenetics (EFI) Conference 2026.... Read more
Prognostic Biomarker Identified in Diffuse Large B-Cell Lymphoma
Diffuse large B-cell lymphoma (DLBCL) is the most common form of non-Hodgkin lymphoma and often presents with aggressive clinical behavior. Although many patients respond to standard chemotherapy with... Read moreImmunology
view channel
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read more
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read morePathology
view channel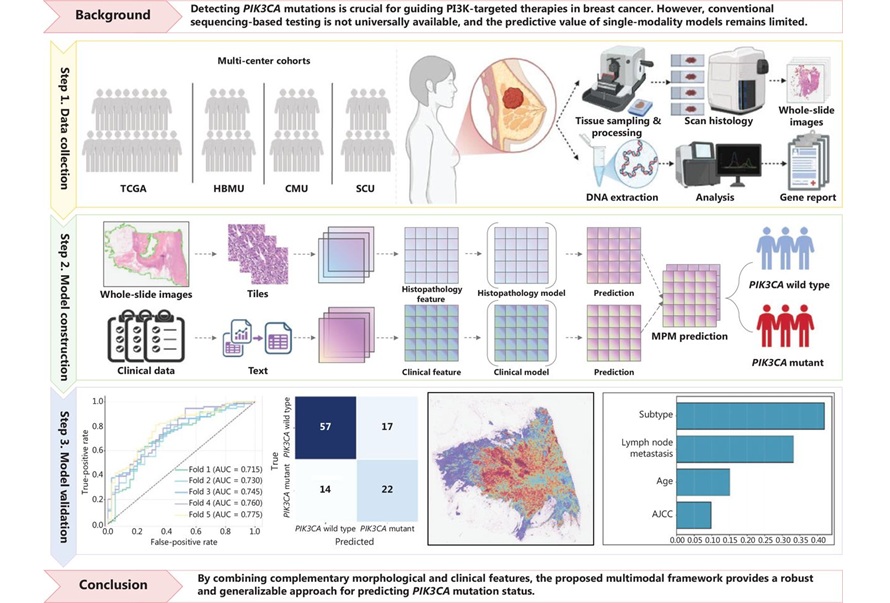
Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
PIK3CA mutations are key biomarkers for selecting phosphoinositide 3-kinase (PI3K)–targeted therapies in breast cancer, yet access to molecular testing can be inconsistent and costly. Conventional polymerase... Read more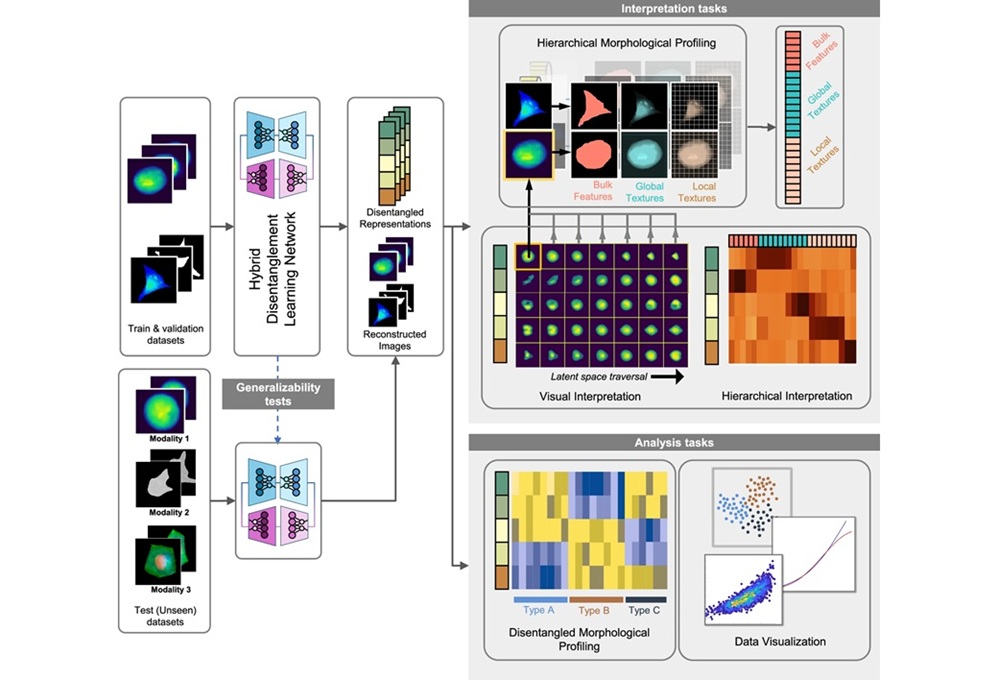
Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
Microscopy images contain rich clues about cell health, but many disease-relevant morphological differences are too subtle to see and difficult to quantify consistently. Artificial intelligence (AI) has... Read moreTechnology
view channel
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read more
Integrated System Streamlines Pre-Analytical Workflow for Molecular Testing
Pre-analytical variation remains a leading source of inconsistent molecular test results and added costs, particularly when laboratories rely on multiple instruments and protocols. Standardizing nucleic... Read moreIndustry
view channel
QuidelOrtho Adds Ultra-Fast PCR Platform with LEX Acquisition
QuidelOrtho Corporation has completed the acquisition of LEX Diagnostics for approximately USD 100 million in cash. The transaction adds the LEX VELO System to QuidelOrtho’s portfolio. The platform received U.... Read more
Seegene Showcases Real-Time PCR Data Analytics Platform at ESCMID
Seegene introduced STAgora, a real-time data analytics platform built on aggregated statistical testing data, at ESCMID Global 2026 in Munich, where it also presented an enhanced model of its automated... Read more
Roche Affiliate Expands MRD Portfolio with SAGA Acquisition
Foundation Medicine, Inc., an independent affiliate of Roche, announced plans to expand its monitoring portfolio with SAGA Diagnostics’ Pathlight, a personalized, tumor-informed molecular residual disease... Read more