Routine Blood Parameters Predict Invasive Aspergillosis Prognosis
|
By LabMedica International staff writers Posted on 19 Nov 2019 |
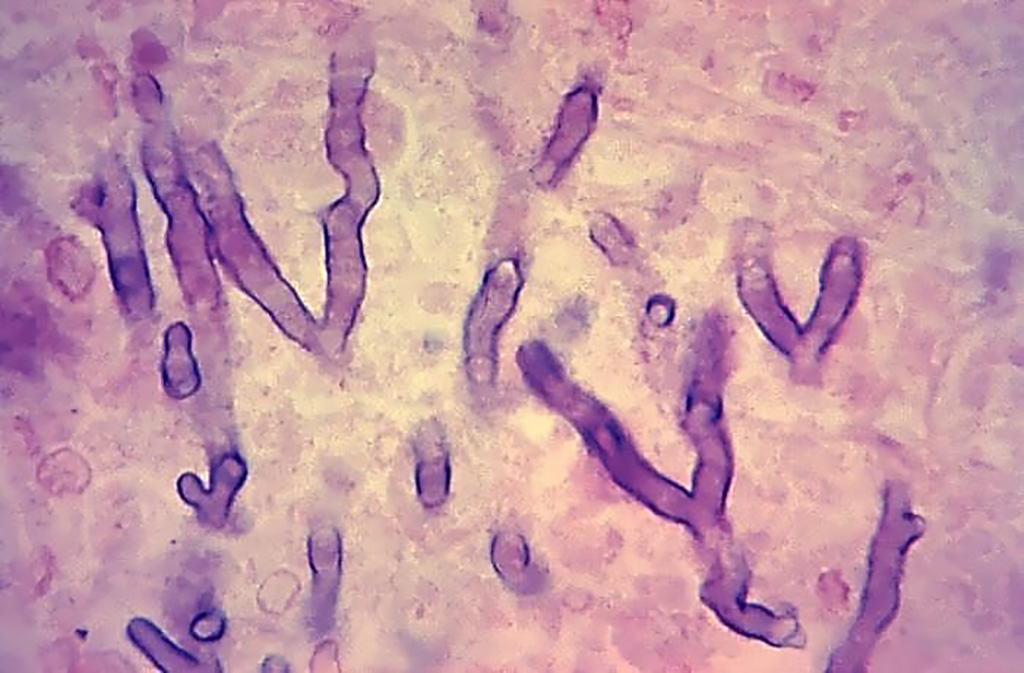
Image: Photomicrograph of a hematoxylin-eosin-stained human lung tissue specimen, harvested from a pulmonary aspergillosis patient. The histopathologic changes indicate the presence of Aspergillus sp. fungal organisms. Note the branching hyphae amongst the lung tissue (Photo courtesy of the Armed Forces Institute of Pathology).
Aspergillosis occurs in chronic or acute forms which are clinically very distinct. Most cases of acute aspergillosis occur in patients with severely compromised immune systems, e.g. those undergoing bone marrow transplantation. Symptoms include fever, chills, shock, delirium, seizures, and blood clots.
Acute invasive aspergillosis occurs when the immune system fails to prevent Aspergillus spores from entering the bloodstream via the lungs. Without the body mounting an effective immune response, fungal cells are free to disseminate throughout the body and can infect major organs such as the heart and kidneys. The patient may develop kidney failure, liver failure (causing jaundice), and breathing difficulties and death can occur quickly.
A large team of scientists working with the National University of Singapore (Singapore) performed a post-hoc secondary analysis of two multicenter randomized trials. The Global Comparative Aspergillosis Study (GCA, N=123) and the Combination Antifungal Study (CAS, N=251) constituted the discovery and validation cohorts respectively. The outcome measures were response to treatment and survival to 12 weeks. Interval platelet, galactomannan index (GMI) and C-reactive protein (CRP) levels prior and during anti-fungal treatment were analyzed.
The investigators reported that the 12-week survival was 70.7% and 63.7% for the GCA and CAS cohorts respectively. In the GCA cohort, every 10×109/L platelet count increase at week 2 and 4 improved 12-week survival odds by 6-18% (Odds ratio [OR] 1.06-1.18). Survival odds also improved 13% with every 10 mg/dL CRP drop at week 1 and 2 (OR 0.87). In the CAS cohort, week 2 platelet count was also associated with 12-week survival with 10% improved odds for every 10×109/L platelet increase (OR, 1.10). A GMI drop of 0.1 units was additionally found to increase the odds of treatment response by 3% at the baseline of week 0 (OR 0.97). Week 2 platelet and CRP levels performed better than GMI on ROC analyses for survival (area under ROC curve 0.76, 0.87 and 0.67 respectively). A baseline platelet count higher than 30×109/L clearly identified patients with > 75% survival probability.
The authors concluded that higher serial platelets were associated with overall survival while GMI trends were linked to invasive aspergillosis treatment response. Routine and simple laboratory indices may aid follow-up of response in invasive aspergillosis patients. The study was published on October 24, 2019 in the journal Clinical Microbiology and Infection.
Related Links:
National University of Singapore
Acute invasive aspergillosis occurs when the immune system fails to prevent Aspergillus spores from entering the bloodstream via the lungs. Without the body mounting an effective immune response, fungal cells are free to disseminate throughout the body and can infect major organs such as the heart and kidneys. The patient may develop kidney failure, liver failure (causing jaundice), and breathing difficulties and death can occur quickly.
A large team of scientists working with the National University of Singapore (Singapore) performed a post-hoc secondary analysis of two multicenter randomized trials. The Global Comparative Aspergillosis Study (GCA, N=123) and the Combination Antifungal Study (CAS, N=251) constituted the discovery and validation cohorts respectively. The outcome measures were response to treatment and survival to 12 weeks. Interval platelet, galactomannan index (GMI) and C-reactive protein (CRP) levels prior and during anti-fungal treatment were analyzed.
The investigators reported that the 12-week survival was 70.7% and 63.7% for the GCA and CAS cohorts respectively. In the GCA cohort, every 10×109/L platelet count increase at week 2 and 4 improved 12-week survival odds by 6-18% (Odds ratio [OR] 1.06-1.18). Survival odds also improved 13% with every 10 mg/dL CRP drop at week 1 and 2 (OR 0.87). In the CAS cohort, week 2 platelet count was also associated with 12-week survival with 10% improved odds for every 10×109/L platelet increase (OR, 1.10). A GMI drop of 0.1 units was additionally found to increase the odds of treatment response by 3% at the baseline of week 0 (OR 0.97). Week 2 platelet and CRP levels performed better than GMI on ROC analyses for survival (area under ROC curve 0.76, 0.87 and 0.67 respectively). A baseline platelet count higher than 30×109/L clearly identified patients with > 75% survival probability.
The authors concluded that higher serial platelets were associated with overall survival while GMI trends were linked to invasive aspergillosis treatment response. Routine and simple laboratory indices may aid follow-up of response in invasive aspergillosis patients. The study was published on October 24, 2019 in the journal Clinical Microbiology and Infection.
Related Links:
National University of Singapore
Latest Microbiology News
- Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
- Rapid Urine Test Speeds Antibiotic Selection for UTIs
- WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
Channels
Clinical Chemistry
view channel
FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
Accurate evaluation of androgen status often requires concordant measurement of total testosterone, free testosterone, and sex hormone‑binding globulin. Reference methods such as equilibrium dialysis with... Read more
CE-Marked Blood Biomarker Test Advances Automated Alzheimer’s Diagnostics
Specialized care settings frequently evaluate patients aged 50 years and older who present with signs and symptoms of cognitive decline to determine whether amyloid pathology linked to Alzheimer’s disease... Read moreMolecular Diagnostics
view channel
Blood-Based “Ageing Clock” Helps Predict Dementia Risk and Earlier Onset
Dementia imposes a growing health burden, affecting an estimated 982,000 people in the UK, with cases projected to reach 1.4 million by 2040. Earlier identification of those most likely to develop disease... Read more
Blood Test Refines Biopsy Decisions in Prostate Cancer
Prostate biopsy decisions remain challenging, with many men undergoing invasive procedures that ultimately yield negative results. In the U.S., more than a million prostate biopsies are performed each... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more