Diagnostic Panel Analysis of Lung Cancer Compared
|
By LabMedica International staff writers Posted on 25 Sep 2019 |

Image: The Quick-Ray Manual Tissue Microarrayer Set (Photo courtesy of Unitma).
Lung cancer is the leading cause of cancer death. Non-small cell lung carcinomas are the most common type of lung cancers. They can further be divided into adenocarcinomas and squamous cell carcinomas. Accurately classifying these tumors is important to select the best treatment options for patients.
Immunohistochemical tests help in improving the histopathological diagnosis of the cancer. However there are many antibodies used in the process and there is a lack of data on panels of antibodies that work best in diagnosing lung carcinomas. The use of appropriate markers for this identification is crucial in order to conserve patient tissue for further molecular testing that could guide treatment decisions and have prognostic implications.
Pathologists at Flinders University (Bedford Park, Australia) selected a cohort of 200 patients, diagnosed with either adenocarcinoma (ADC) or squamous cell carcinomas (SCC) at the Flinders Medical Centre between 1991 and 2011. All patients had histological diagnosis on resection specimens and cases were reclassified according to the 2015 WHO classification of tumors of the lung. These cases had initially been diagnosed based on morphology, immunohistochemistry (IHC) and electron microscopy.
Tissue cylinders with a diameter of 2.0 mm were punched from representative tumor regions of each donor tissue block and brought into recipient paraffin block using a Quick-Ray Manual Tissue Microarrayer. Control tissue was included in each tissue microarray (TMA). There were 116 SCC and 84 ADC. The team performed IHC on a Ventana BenchMark ULTRA immunostainer with three TTF-1 clones (SP141, SPT24 and 8G7G3/1), Napsin A, p40, p63 and CK5/6 and panels of four or two markers that best help identify adenocarcinoma and squamous cell carcinoma were ascertained.
The scientists reported that their results showed that the best four-marker panel utilized TTF-1 (clone SP141), Napsin A, p63 and CK5/6 with a sensitivity of 98.3% and high specificity of 91.7%. The best two-marker panel was TTF-1 (clone SP141) and p63 with 96.5% sensitivity and 85.71% specificity. The authors concluded that as there are variations in the performance of different clones of TTF-1 IHC antibodies, the clone chosen can increase the diagnostic value in differentiating adenocarcinoma from squamous cell carcinoma. In the panels analyzed, the survival of cases concordant with the diagnosis had longer survival compared to those that were discordant. The difference was however not statistically significant. The study was published on September 11, 2019, in the journal Pathology and Laboratory Medicine International.
Related Links:
Flinders University
Immunohistochemical tests help in improving the histopathological diagnosis of the cancer. However there are many antibodies used in the process and there is a lack of data on panels of antibodies that work best in diagnosing lung carcinomas. The use of appropriate markers for this identification is crucial in order to conserve patient tissue for further molecular testing that could guide treatment decisions and have prognostic implications.
Pathologists at Flinders University (Bedford Park, Australia) selected a cohort of 200 patients, diagnosed with either adenocarcinoma (ADC) or squamous cell carcinomas (SCC) at the Flinders Medical Centre between 1991 and 2011. All patients had histological diagnosis on resection specimens and cases were reclassified according to the 2015 WHO classification of tumors of the lung. These cases had initially been diagnosed based on morphology, immunohistochemistry (IHC) and electron microscopy.
Tissue cylinders with a diameter of 2.0 mm were punched from representative tumor regions of each donor tissue block and brought into recipient paraffin block using a Quick-Ray Manual Tissue Microarrayer. Control tissue was included in each tissue microarray (TMA). There were 116 SCC and 84 ADC. The team performed IHC on a Ventana BenchMark ULTRA immunostainer with three TTF-1 clones (SP141, SPT24 and 8G7G3/1), Napsin A, p40, p63 and CK5/6 and panels of four or two markers that best help identify adenocarcinoma and squamous cell carcinoma were ascertained.
The scientists reported that their results showed that the best four-marker panel utilized TTF-1 (clone SP141), Napsin A, p63 and CK5/6 with a sensitivity of 98.3% and high specificity of 91.7%. The best two-marker panel was TTF-1 (clone SP141) and p63 with 96.5% sensitivity and 85.71% specificity. The authors concluded that as there are variations in the performance of different clones of TTF-1 IHC antibodies, the clone chosen can increase the diagnostic value in differentiating adenocarcinoma from squamous cell carcinoma. In the panels analyzed, the survival of cases concordant with the diagnosis had longer survival compared to those that were discordant. The difference was however not statistically significant. The study was published on September 11, 2019, in the journal Pathology and Laboratory Medicine International.
Related Links:
Flinders University
Latest Pathology News
- AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
- FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
- New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
- AI System Analyzes Routine Pathology Slides to Predict Cancer Outcomes
- New Tissue Mapping Approach Identifies High-Risk Form of Diabetic Kidney Disease
- Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
- Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
- Tumor Immune Structure Predicts Response to Immunotherapy in Melanoma
- Plug-and-Play AI Pathology System Classifies Multiple Cancers from Few Slides
- AI-Based Assays Support Risk Stratification in Prostate and Breast Cancer
- AI Pathology Model Predicts Immunotherapy Response in Lung Cancer
- Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
- AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
- Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
- Biomarker Predicts Immunotherapy Response and Prognosis in Colorectal Cancer
- AI Improves Completeness of Complex Cancer Pathology Reports
Channels
Clinical Chemistry
view channel
Urine-Based Nanosensor Tracks Lung Cancer and Fibrosis Noninvasively
Lung cancer remains difficult to monitor for early progression and treatment resistance, while pulmonary fibrosis continues to pose major challenges for early diagnosis. Clinicians need repeatable, noninvasive... Read more
Blood-Based Alzheimer’s Test Gains CE Mark for Amyloid Pathology Detection
Alzheimer’s disease is the most common cause of dementia, yet confirmatory testing remains invasive and hard to access. Diagnosis currently takes an average of 3.5 years, and about 75% of people with dementia... Read moreMolecular Diagnostics
view channel
Tumor Genomic Profiling Identifies High-Risk Gallbladder Cancer
Gallbladder cancer is an aggressive biliary tract malignancy that often progresses silently and is frequently diagnosed at an advanced stage. Five-year survival is often below 10%, and only an estimated... Read more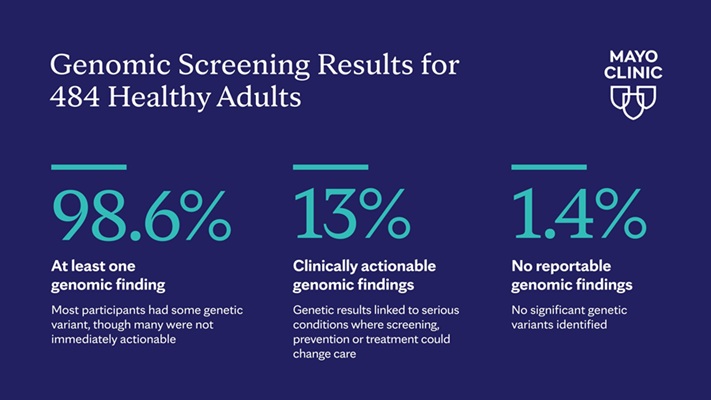
Genome Sequencing Uncovers Hidden Genetic Risks in Healthy Adults
Genetic risk for serious diseases can go undetected in routine care, limiting opportunities for earlier intervention. Translating genomic data into actionable care plans also creates operational challenges... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read more
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read moreMicrobiology
view channel
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read moreTechnology
view channel
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read more
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read moreIndustry
view channel
AI-Powered Multi-Functional Analyzer Wins German Innovation Award
Hematology services are increasingly delivered across distributed care settings, where limited staffing and complex workflows can extend turnaround times. Advanced morphology review still often depends... Read more
























