New Automated Assay Measures Levels of sPLA2-IIA
|
By LabMedica International staff writers Posted on 08 Apr 2019 |
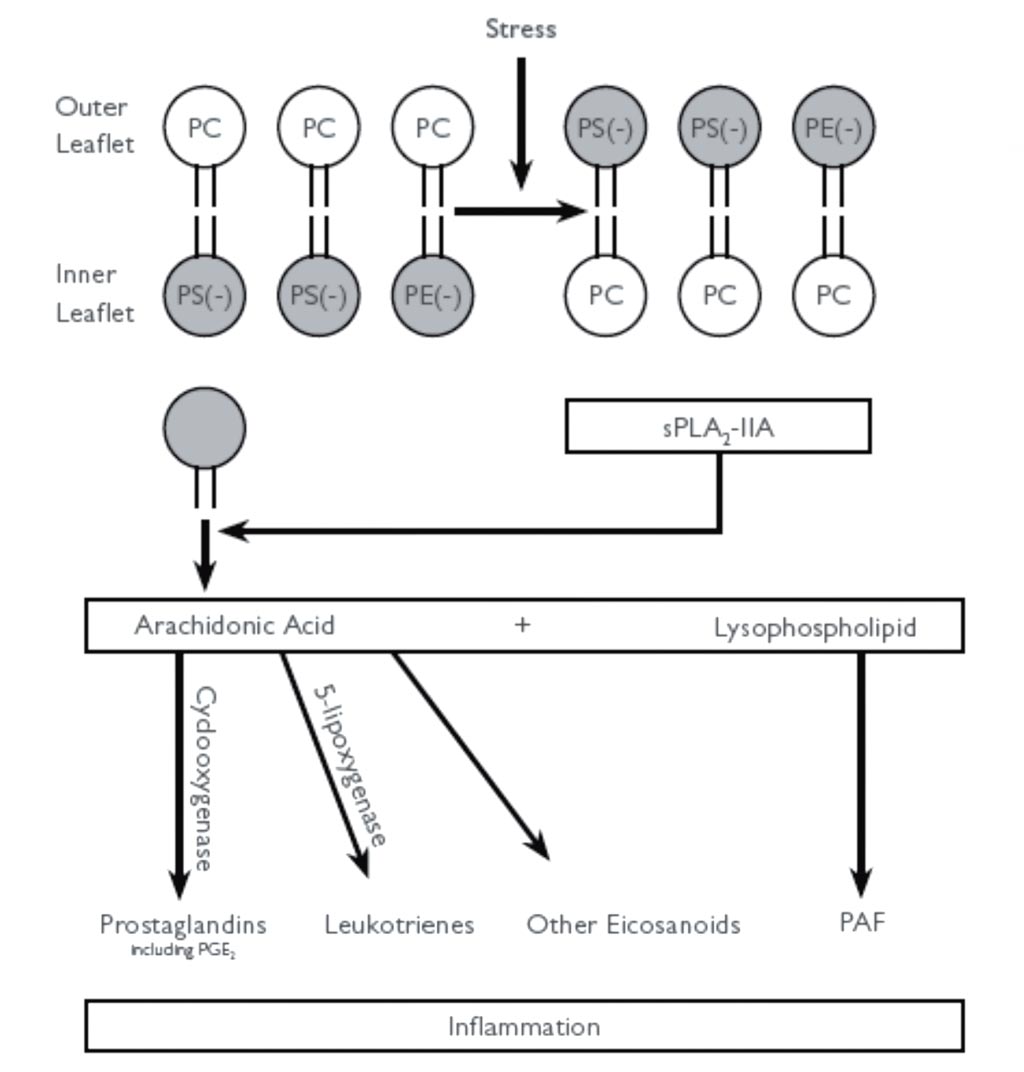
Image: The diagram shows how sPLA2-IIA binding to anionic phospholipids leads to its activation and promotes inflammation. The oxidation of the phospholipids produces stress, which causes the anionic phospholipids phosphatidylserine (PS) and phosphatidylethanolamine (PE) to be transported to the outer leaflet. This reaction activates cationic sPLA2-IIA. The higher activity of sPLA2-IIA promotes the hydrolysis of the outer leaflet phospholipids into arachidonic acid and lysophospholipids. Through the cyclooxygenase and 5-lipoxygenase enzymes, arachidonic acid is converted into prostaglandins, leukotrienes, and other inflammatory eicosanoids (Photo courtesy of Randox).
Phospholipase A2 (PLA2s) are a family of enzymes that cleave the fatty acid in position two of phospholipids, hydrolyzing the bond between the second fatty acid “tail” and the glycerol molecule. Of particular interest is the secreted phospholipase (sPLA2-IIA) that specifically recognizes the sn-2 acyl bond of phospholipids and catalytically hydrolyzes the bond, releasing arachidonic acid and lysophosphatidic acid. Upon downstream modification by cyclooxygenases, arachidonic acid is modified into active eicosanoid compounds, including prostaglandins and leukotrienes.
sPLA2-IIA has been shown to promote inflammation in mammals by catalyzing the first step of the arachidonic acid pathway by breaking down phospholipids, resulting in the formation of fatty acids including arachidonic acid. This arachidonic acid is then metabolized to form several inflammatory and thrombogenic molecules. Elevated levels of sPLA2-IIA are thought to contribute to several inflammatory diseases, and have been shown to promote vascular inflammation correlating with disease risk in coronary artery disease and acute coronary syndrome. Unlike traditional cardiac biomarkers used to predict adverse outcomes in patients, sPLA2-IIA has been shown to act at multiple pathways involved in atherogenesis, from lipid oxidation to modulation of vascular inflammatory cell activation and apoptosis. Measurement of sPLA2-IIA enables clinicians to gain a comprehensive overview of cardiac risk and may help to tailor treatment accordingly.
Recognizing the link between the secreted phospholipases and coronary disease risk, Randox (Crumlin, United Kingdom) has introduced an automated assay (currently for research use only) to measure levels of sPLA2-IIA. This automated assay eliminates the inconvenient and time consuming traditional ELISA based test for sPLA2-IIA.
The Randox sPLA2-IIA Assay was designed for use on a wide range of clinical chemistry analysers, and applications are available detailing instrument-specific settings. The latex enhanced immunoturbidimetric assay kit contains liquid ready-to-use reagents for convenience and ease-of-use. In addition, controls and calibrators are available offering a complete testing package. The addition of the sPLA2-IIA Assay compliments the existing cardiac risk panel from Randox, providing a different outlook and method of assessing cardiac concerns in patients.
sPLA2-IIA has been shown to promote inflammation in mammals by catalyzing the first step of the arachidonic acid pathway by breaking down phospholipids, resulting in the formation of fatty acids including arachidonic acid. This arachidonic acid is then metabolized to form several inflammatory and thrombogenic molecules. Elevated levels of sPLA2-IIA are thought to contribute to several inflammatory diseases, and have been shown to promote vascular inflammation correlating with disease risk in coronary artery disease and acute coronary syndrome. Unlike traditional cardiac biomarkers used to predict adverse outcomes in patients, sPLA2-IIA has been shown to act at multiple pathways involved in atherogenesis, from lipid oxidation to modulation of vascular inflammatory cell activation and apoptosis. Measurement of sPLA2-IIA enables clinicians to gain a comprehensive overview of cardiac risk and may help to tailor treatment accordingly.
Recognizing the link between the secreted phospholipases and coronary disease risk, Randox (Crumlin, United Kingdom) has introduced an automated assay (currently for research use only) to measure levels of sPLA2-IIA. This automated assay eliminates the inconvenient and time consuming traditional ELISA based test for sPLA2-IIA.
The Randox sPLA2-IIA Assay was designed for use on a wide range of clinical chemistry analysers, and applications are available detailing instrument-specific settings. The latex enhanced immunoturbidimetric assay kit contains liquid ready-to-use reagents for convenience and ease-of-use. In addition, controls and calibrators are available offering a complete testing package. The addition of the sPLA2-IIA Assay compliments the existing cardiac risk panel from Randox, providing a different outlook and method of assessing cardiac concerns in patients.
Latest Clinical Chem. News
- FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
- CE-Marked Blood Biomarker Test Advances Automated Alzheimer’s Diagnostics
- Noninvasive Urine Test May Support Earlier Diagnosis of Psychiatric Disorders
- At-Home Blood and Cognitive Tests Support Dementia Risk Stratification
- Ultrasensitive Test Detects Key Biomarker of Frontotemporal Dementia Subtype
- Routine Blood Tests Years Before Pregnancy Could Identify Preeclampsia Risk
- Blood Test Detects Testicular Cancer Missed by Standard Markers
- Routine Blood Tests Identify Biomarkers Linked to PTSD
- Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
- Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
- Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
- Rapid Biosensor Detects Pancreatic Cancer Biomarker for Early Detection
- Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
- Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
- Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
- AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Channels
Molecular Diagnostics
view channel
Blood-Based “Ageing Clock” Helps Predict Dementia Risk and Earlier Onset
Dementia imposes a growing health burden, affecting an estimated 982,000 people in the UK, with cases projected to reach 1.4 million by 2040. Earlier identification of those most likely to develop disease... Read more
Blood Test Refines Biopsy Decisions in Prostate Cancer
Prostate biopsy decisions remain challenging, with many men undergoing invasive procedures that ultimately yield negative results. In the U.S., more than a million prostate biopsies are performed each... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read more
Point-of-Care Tests Could Expand Access to Mpox Diagnosis
Mpox outbreaks in non-endemic regions have underscored the need for rapid, accessible diagnostics to limit transmission. Polymerase chain reaction (PCR) remains the clinical reference, yet it depends on... Read moreMicrobiology
view channel
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read more
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read morePathology
view channel
FDA Clears AI Digital Pathology Tool for Breast Cancer Risk Stratification
Risk assessment at diagnosis is central to guiding therapy for early-stage, hormone receptor-positive, human epidermal growth factor receptor 2-negative (HR+/HER2-) invasive breast cancer, where overtreatment... Read more
New AI Tool Reveals Hidden Genetic Signals in Routine H&E Slides
Pathologists worldwide rely on hematoxylin and eosin (H&E) slides to examine tissue architecture, yet these stains do not reveal the underlying molecular activity that often drives disease.... Read moreTechnology
view channel
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read more
Tumor-on-a-Chip Platform Models Pancreatic Cancer Treatment Response
Pancreatic cancer remains one of the hardest malignancies to treat because tumors are embedded within a dense microenvironment that shapes growth and therapy response. Standard laboratory models often... Read moreIndustry
view channel
Roche to Acquire PathAI for Up to $1.05 Billion to Strengthen AI Diagnostics Portfolio
Roche has entered into a definitive merger agreement to acquire PathAI, a company focused on digital pathology and artificial intelligence for pathology laboratories and the biopharma industry.... Read more