Nanopatterned Microfluidic Chip Detects Cancer Markers
|
By LabMedica International staff writers Posted on 13 Mar 2019 |
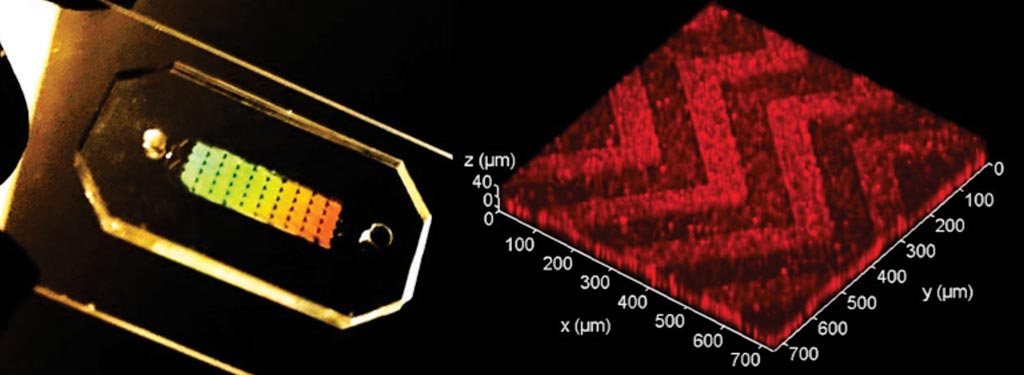
Image: The three-dimensional (3D) herringbone nanopatterned microfluidic chip that detects cancer faster (Photo courtesy of the University of Kansas).
The performance of current microfluidic methods for exosome detection is constrained by boundary conditions, as well as fundamental limits to microscale mass transfer and interfacial exosome binding.
Some types of cancer, such as ovarian cancer, tend to remain undetected until they are too advanced for treatment to be effective. Now, an innovative tool may be able to detect cancer easily, quickly, and in minuscule amounts of blood. A device, which is called a "3D-nanopatterned microfluidic chip," could successfully detect cancer markers in the tiniest drop of blood or in a component of the blood called plasma.
Biochemists at the University of Kansas (Lawrence, KS, USA) and their colleagues have shown that a microfluidic chip designed with self-assembled three-dimensional herringbone nanopatterns can detect low levels of tumor-associated exosomes in plasma (10 exosomes μL−1, or approximately 200 vesicles per 20 μL of spiked sample) that would otherwise be undetectable by standard microfluidic systems for biosensing. The nanopatterns promote microscale mass transfer, increase surface area and probe density to enhance the efficiency and speed of exosome binding, and permit drainage of the boundary fluid to reduce near-surface hydrodynamic resistance, thus promoting particle–surface interactions for exosome binding.
The scientists used the device for the detection, in 2 μL plasma samples from 20 ovarian cancer patients and 10 age-matched controls, of exosome subpopulations expressing CD24, epithelial cell adhesion molecule and folate receptor alpha proteins. They suggest exosomal folate receptor alpha as a potential biomarker for early detection and progression monitoring of ovarian cancer. The nanolithography-free nanopatterned device should facilitate the use of liquid biopsies for cancer diagnosis.
Yong Zeng, PhD, an associate professor of chemistry and the senior author of the study, said, “Historically, people thought exosomes were like 'trash bags' that cells could use to dump unwanted cellular contents. But, in the past decade scientists realized they were quite useful for sending messages to recipient cells and communicating molecular information important in many biological functions. Basically, tumors send out exosomes packaging active molecules that mirror the biological features of the parental cells. While all cells produce exosomes, tumor cells are really active compared to normal cells.” The study was published on February 25, 2019, in the journal Nature Biomedical Engineering.
Related Links:
University of Kansas
Some types of cancer, such as ovarian cancer, tend to remain undetected until they are too advanced for treatment to be effective. Now, an innovative tool may be able to detect cancer easily, quickly, and in minuscule amounts of blood. A device, which is called a "3D-nanopatterned microfluidic chip," could successfully detect cancer markers in the tiniest drop of blood or in a component of the blood called plasma.
Biochemists at the University of Kansas (Lawrence, KS, USA) and their colleagues have shown that a microfluidic chip designed with self-assembled three-dimensional herringbone nanopatterns can detect low levels of tumor-associated exosomes in plasma (10 exosomes μL−1, or approximately 200 vesicles per 20 μL of spiked sample) that would otherwise be undetectable by standard microfluidic systems for biosensing. The nanopatterns promote microscale mass transfer, increase surface area and probe density to enhance the efficiency and speed of exosome binding, and permit drainage of the boundary fluid to reduce near-surface hydrodynamic resistance, thus promoting particle–surface interactions for exosome binding.
The scientists used the device for the detection, in 2 μL plasma samples from 20 ovarian cancer patients and 10 age-matched controls, of exosome subpopulations expressing CD24, epithelial cell adhesion molecule and folate receptor alpha proteins. They suggest exosomal folate receptor alpha as a potential biomarker for early detection and progression monitoring of ovarian cancer. The nanolithography-free nanopatterned device should facilitate the use of liquid biopsies for cancer diagnosis.
Yong Zeng, PhD, an associate professor of chemistry and the senior author of the study, said, “Historically, people thought exosomes were like 'trash bags' that cells could use to dump unwanted cellular contents. But, in the past decade scientists realized they were quite useful for sending messages to recipient cells and communicating molecular information important in many biological functions. Basically, tumors send out exosomes packaging active molecules that mirror the biological features of the parental cells. While all cells produce exosomes, tumor cells are really active compared to normal cells.” The study was published on February 25, 2019, in the journal Nature Biomedical Engineering.
Related Links:
University of Kansas
Latest Clinical Chem. News
- Automated NfL Assay Supports Monitoring of Neurological Disorders
- Blood-Based Screening Test Targets Early Detection of Colorectal Cancer
- New CLIA Status Brings Mass Spectrometry Steroid Testing to Routine Labs
- CSF Biomarker Improves Diagnosis of Parkinson’s Disease and Lewy Body Dementia
- Simple Urine Home Test Kit Could Detect Early-Stage Breast Cancer
- Study Shows Dual Biomarkers Improve Accuracy of Alzheimer’s Detection
- New Tool Tracks Biomarker Changes to Predict Myeloma Progression
- New Plasma Tau Assay Improves Prediction of Alzheimer’s Progression
- First IVD Immunoassay to Detect Alzheimer’s Risk Gene Variant Receives CE Mark
- Routine Blood Markers Predict Heart Failure Risk in Prediabetes
- AI Model Enables Personalized Glucose Predictions for Type 1 Diabetes
- AI-Powered Blood Test Distinguishes Deadly Cardiac Events
- AI Sensor Detects Neurological Disorders Using Single Saliva Drop
- Blood Test Tracks Transplant Health Using Donor DNA
- New Blood Test Index Offers Earlier Detection of Liver Scarring
- Electronic Nose Smells Early Signs of Ovarian Cancer in Blood
Channels
Molecular Diagnostics
view channel
First IVDR‑Certified IGH Clonality Assay Supports Diagnosis of B-Cell Malignancies
Accurate identification of clonal immunoglobulin heavy chain (IGH) gene rearrangements is central to evaluating suspected B-cell lymphoproliferative disorders, where a single B-cell clone yields a defining... Read more
Plasma ctDNA Testing Predicts Breast Cancer Recurrence After Neoadjuvant Therapy
Accurate identification of breast cancer patients at risk of relapse after pre-surgery treatment is central to guiding adjuvant decisions, particularly in aggressive disease. Circulating fragments of tumor... Read moreHematology
view channel
Rapid Cartridge-Based Test Aims to Expand Access to Hemoglobin Disorder Diagnosis
Sickle cell disease and beta thalassemia are hemoglobin disorders that often require referral to specialized laboratories for definitive diagnosis, delaying results for patients and clinicians.... Read more
New Guidelines Aim to Improve AL Amyloidosis Diagnosis
Light chain (AL) amyloidosis is a rare, life-threatening bone marrow disorder in which abnormal amyloid proteins accumulate in organs. Approximately 3,260 people in the United States are diagnosed... Read moreImmunology
view channel
FDA Approval Expands Use of PD-L1 Companion Diagnostic in Esophageal and GEJ Carcinomas
Esophageal and gastroesophageal junction carcinomas (GEJ) have a poor prognosis, with approximately 16,250 deaths in the United States in 2025 and a five-year relative survival of 21.9%.... Read more
Study Identifies Inflammatory Pathway Driving Immunotherapy Resistance in Bladder Cancer
Bladder cancer remains a prevalent malignancy with variable responses to immune checkpoint inhibitors. Clinicians often observe elevated C-reactive protein and interleukin-6 in affected patients, yet the... Read moreMicrobiology
view channel
WHO Endorses Rapid Point-of-Care Testing to Improve TB Detection
Tuberculosis (TB) remains a leading infectious killer, with more than 3,300 deaths and 29,000 new illnesses every day. Diagnostic delays and dependence on centralized laboratory networks continue to impede... Read more
Breath Analysis Approach Offers Rapid Detection of Bacterial Infection
Accurate and rapid identification of bacterial infections remains challenging in acute care, where delays can hinder timely, targeted therapy. Infectious diseases are a major cause of mortality worldwide,... Read more
Study Highlights Accuracy Gaps in Consumer Gut Microbiome Kits
Direct-to-consumer gut microbiome kits promise personalized insights by profiling fecal bacteria and generating health readouts, but their analytical accuracy remains uncertain. A new study shows that... Read more
WHO Recommends Near POC Tests, Tongue Swabs and Sputum Pooling for TB Diagnosis
Tuberculosis (TB) remains one of the world’s leading infectious disease killers, yet millions of cases go undiagnosed or are detected too late. Barriers such as reliance on sputum samples, limited laboratory... Read morePathology
view channel
Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Lung cancer is the leading cause of cancer death, killing more people in the United States than breast, prostate, and colon cancers combined. In lung adenocarcinoma (LUAD), tumors that invade nearby blood... Read more
AI-Powered Tool to Transform Dermatopathology Workflow
Skin cancer accounts for the largest number of cancer diagnoses in the United States, placing sustained pressure on pathology services. Diagnostic interpretation can be variable for challenging melanocytic... Read moreTechnology
view channel
Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
Chemotherapy selection for breast cancer is challenged by heterogeneous tumor responses. Conventional chemosensitivity assays can be slow, require large sample volumes, and struggle with complex biological... Read more
Online Tool Supports Family Screening for Inherited Cancer Risk
Genetic test results in oncology often have implications for relatives who may share inherited cancer risk. Many health systems lack structured processes to help patients alert family members, limiting... Read moreIndustry
view channel
Integrated DNA Technologies Expands into Clinical Diagnostics
Integrated DNA Technologies (IDT; Coralville, Iowa, USA) has announced the launch of Archer FUSIONPlex-HT Dx and VARIANTPlex-HT Dx. This launch marks the company’s first in vitro diagnostic (IVD) offerings... Read more