Levels of Bim Protein in T-cells Reflect Success of Anti–PD-1 Cancer Therapy
|
By LabMedica International staff writers Posted on 16 May 2016 |
Image: A structural model of the Bim protein (Photo courtesy of Wikimedia Commons).
Measurement of levels of Bim (BCL-2-interacting mediator of cell death) protein in circulating T-cells of cancer patients may provide a less invasive strategy to predict and monitor responses to anti–PD-1 therapy.
Immune checkpoint therapy with PD-1 (Programmed cell death protein 1) blockade has emerged as an effective therapy for many advanced cancers; however, only a small fraction of patients achieve long-term responses. There is no validated blood-based means of predicting the response to PD-1 blockade.
PD-1, functioning as an immune checkpoint, plays an important role in down regulating the immune system by preventing the activation of T-cells, which in turn reduces autoimmunity and promotes self-tolerance. The inhibitory effect of PD-1 is accomplished through a dual mechanism of promoting apoptosis (programmed cell death) in antigen specific T-cells in lymph nodes while simultaneously reducing apoptosis in regulatory T cells (suppressor T cells).
Investigators at the Mayo Clinic (Rochester, MN, USA) had previously cloned PD-L1 (Programmed death-ligand 1) and found that tumor-associated PD-L1 mediated tumor immune evasion. Since then the group has been working on dissecting the molecular mechanisms of the PD-L1/PD-1 pathway in T-cell dysfunction.
They recently reported that they had identified the protein Bim as a downstream signaling molecule of the PD-1 pathway and that its detection in T-cells was significantly associated with expression of PD-1 and effector T-cell markers. Thus, high levels of Bim in circulating tumor-reactive T-cells were prognostic of poor survival in patients with metastatic melanoma who did not receive anti–PD-1 therapy and were also predictive of clinical benefit in patients with metastatic melanoma who received anti–PD-1 therapy in the form of the humanized monoclonal antibody drug pembrolizumab. This circulating tumor-reactive T-cell population significantly decreased after successful anti–PD-1 therapy.
"Our previous research demonstrated that Bim is a downstream signaling molecule in the PD-1 signaling pathway, and that levels of Bim reflect the degree of PD-1 interaction with its ligand PD-L1," said senior author Dr. Haidong Dong, associate professor of immunology at the Mayo Clinic. "We hypothesized that the increased frequency of CD8+PD-1+Bim+T cells in patients who respond to immunotherapy reflects an increased number of target T-cells for PD-1 blockade with pembrolizumab, which may explain the positive clinical outcomes in these patients."
The study was published in the May 5, 2016, online edition of the journal JCI Insight.
Related Links:
Mayo Clinic
Immune checkpoint therapy with PD-1 (Programmed cell death protein 1) blockade has emerged as an effective therapy for many advanced cancers; however, only a small fraction of patients achieve long-term responses. There is no validated blood-based means of predicting the response to PD-1 blockade.
PD-1, functioning as an immune checkpoint, plays an important role in down regulating the immune system by preventing the activation of T-cells, which in turn reduces autoimmunity and promotes self-tolerance. The inhibitory effect of PD-1 is accomplished through a dual mechanism of promoting apoptosis (programmed cell death) in antigen specific T-cells in lymph nodes while simultaneously reducing apoptosis in regulatory T cells (suppressor T cells).
Investigators at the Mayo Clinic (Rochester, MN, USA) had previously cloned PD-L1 (Programmed death-ligand 1) and found that tumor-associated PD-L1 mediated tumor immune evasion. Since then the group has been working on dissecting the molecular mechanisms of the PD-L1/PD-1 pathway in T-cell dysfunction.
They recently reported that they had identified the protein Bim as a downstream signaling molecule of the PD-1 pathway and that its detection in T-cells was significantly associated with expression of PD-1 and effector T-cell markers. Thus, high levels of Bim in circulating tumor-reactive T-cells were prognostic of poor survival in patients with metastatic melanoma who did not receive anti–PD-1 therapy and were also predictive of clinical benefit in patients with metastatic melanoma who received anti–PD-1 therapy in the form of the humanized monoclonal antibody drug pembrolizumab. This circulating tumor-reactive T-cell population significantly decreased after successful anti–PD-1 therapy.
"Our previous research demonstrated that Bim is a downstream signaling molecule in the PD-1 signaling pathway, and that levels of Bim reflect the degree of PD-1 interaction with its ligand PD-L1," said senior author Dr. Haidong Dong, associate professor of immunology at the Mayo Clinic. "We hypothesized that the increased frequency of CD8+PD-1+Bim+T cells in patients who respond to immunotherapy reflects an increased number of target T-cells for PD-1 blockade with pembrolizumab, which may explain the positive clinical outcomes in these patients."
The study was published in the May 5, 2016, online edition of the journal JCI Insight.
Related Links:
Mayo Clinic
Latest Pathology News
- Multimodal AI Tool Predicts Genetic Alterations to Guide Breast Cancer Treatment
- Interpretable AI Reveals Hidden Cellular Features from Microscopy Images
- Tumor Immune Structure Predicts Response to Immunotherapy in Melanoma
- Plug-and-Play AI Pathology System Classifies Multiple Cancers from Few Slides
- AI-Based Assays Support Risk Stratification in Prostate and Breast Cancer
- AI Pathology Model Predicts Immunotherapy Response in Lung Cancer
- Study Reveals Moleclar Mechanism Driving Aggressive Skin Cancer
- AI Precision Tests Deliver Cancer Risk Insights from Routine H&E Slides
- Collaboration Applies AI Pathology to Predict Response to Antibody-Drug Conjugates
- Biomarker Predicts Immunotherapy Response and Prognosis in Colorectal Cancer
- AI Improves Completeness of Complex Cancer Pathology Reports
- AI Tool Predicts Chemotherapy Response in Small Cell Lung Cancer
- Tumor-Specific Biomarker Predicts Neoadjuvant Immunotherapy Response in Gastric Cancer
- AI Tool Predicts Patient-Specific Chemotherapy Benefit in Breast Cancer
- AI-Based Pathology Model Guides Chemotherapy Decisions in Breast Cancer
- Biopsy-Based Gene Test Predicts Recurrence Risk in Lung Adenocarcinoma
Channels
Clinical Chemistry
view channel
Blood Test Detects Testicular Cancer Missed by Standard Markers
Testicular cancer most often affects adolescents and young adults and is highly treatable when found early. Diagnosis can be difficult when tumors do not produce sufficient levels of standard blood-based... Read more
Routine Blood Tests Identify Biomarkers Linked to PTSD
Post-traumatic stress disorder (PTSD) is associated with a range of chronic physical health conditions and affects multiple organ systems. Clinical laboratories routinely measure blood analytes that reflect... Read moreMolecular Diagnostics
view channel
Proteomic Risk Score Predicts Kidney Disease Progression in High-Risk Patients
Chronic kidney disease progression is difficult to anticipate, limiting opportunities to intervene before irreversible decline. Risk assessment is especially uncertain for people who carry high‑risk variants... Read more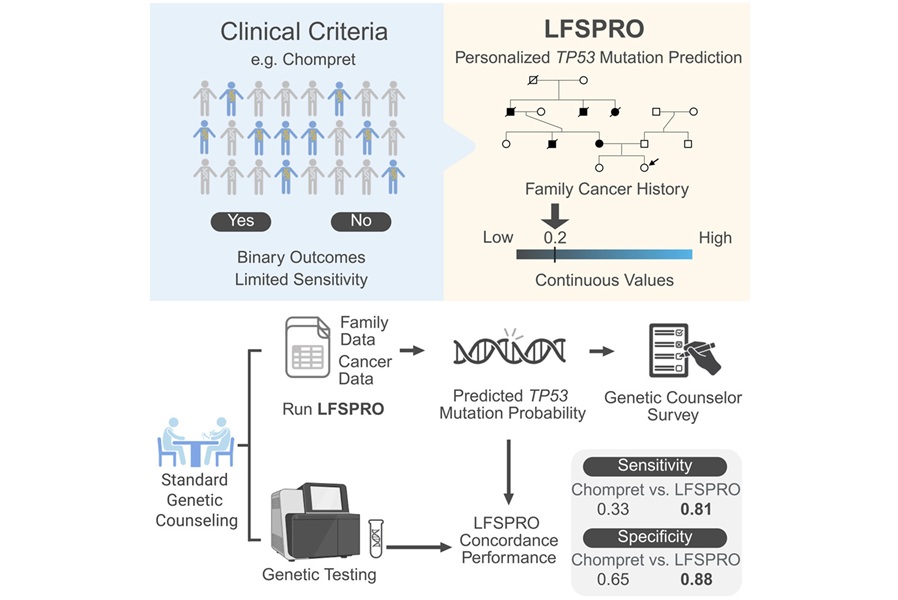
Risk Prediction Tool Enhances Genetic Testing for Li-Fraumeni Syndrome
Li-Fraumeni syndrome is a rare hereditary cancer predisposition most often driven by germline mutations in the TP53 tumor suppressor gene. Determining who should receive TP53 testing remains challenging... Read moreHematology
view channel
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read more
Blood Test Enables Early Detection of Multiple Myeloma Relapse
Bone marrow biopsies remain central to diagnosing and monitoring multiple myeloma, yet the procedure is painful, invasive, and often repeated over time. Older patients—who represent most new cases—can... Read moreImmunology
view channel
Study Highlights Low Sensitivity of Current Lyme Tests in Early Infection
Accurate laboratory diagnosis of early Lyme disease remains challenging because serologic responses may be limited soon after infection. Missed detection at this stage can delay evaluation and management... Read more
Immune Aging Clock Quantifies Immunosenescence and Identifies Therapeutic Target
Immune aging undermines host defense and contributes to multiple age-related diseases, yet its heterogeneity complicates measurement and intervention. Clinical laboratories increasingly seek objective... Read moreMicrobiology
view channel
Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
Tuberculosis remains a major global health challenge and continues to drive significant morbidity and mortality. The World Health Organization’s 2024 global report cites it as the leading cause of death... Read more
Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
Early detection of gastric cancer could be advanced by scalable screening strategies using minimally invasive sampling. Saliva collection is noninvasive and cost-effective, supporting wider adoption... Read moreTechnology
view channel
Microfluidic Single-Cell Assay Predicts Breast Cancer Risk
Risk stratification for breast cancer remains imprecise, as population-based models and breast density can over- or underestimate individual risk, potentially leading to over- or under-screening.... Read more
AI Tool Predicts Non-Response to Targeted Therapy in Colorectal Cancer
Advanced bowel cancer remains difficult to treat, and many patients receive targeted therapies that do not help them but still cause harm. Clinicians need reliable ways to identify likely responders before... Read moreIndustry
view channel
Thermo Fisher Scientific to Sell Microbiology Business to Astorg
Thermo Fisher Scientific has signed a definitive agreement to sell its microbiology business to Astorg for approximately $1.075 billion, comprising cash and a $50 million seller note. Announced on April... Read more