Immunoassay Detects Severe Fever with Thrombocytopenia Syndrome Virus
|
By LabMedica International staff writers Posted on 25 Apr 2016 |
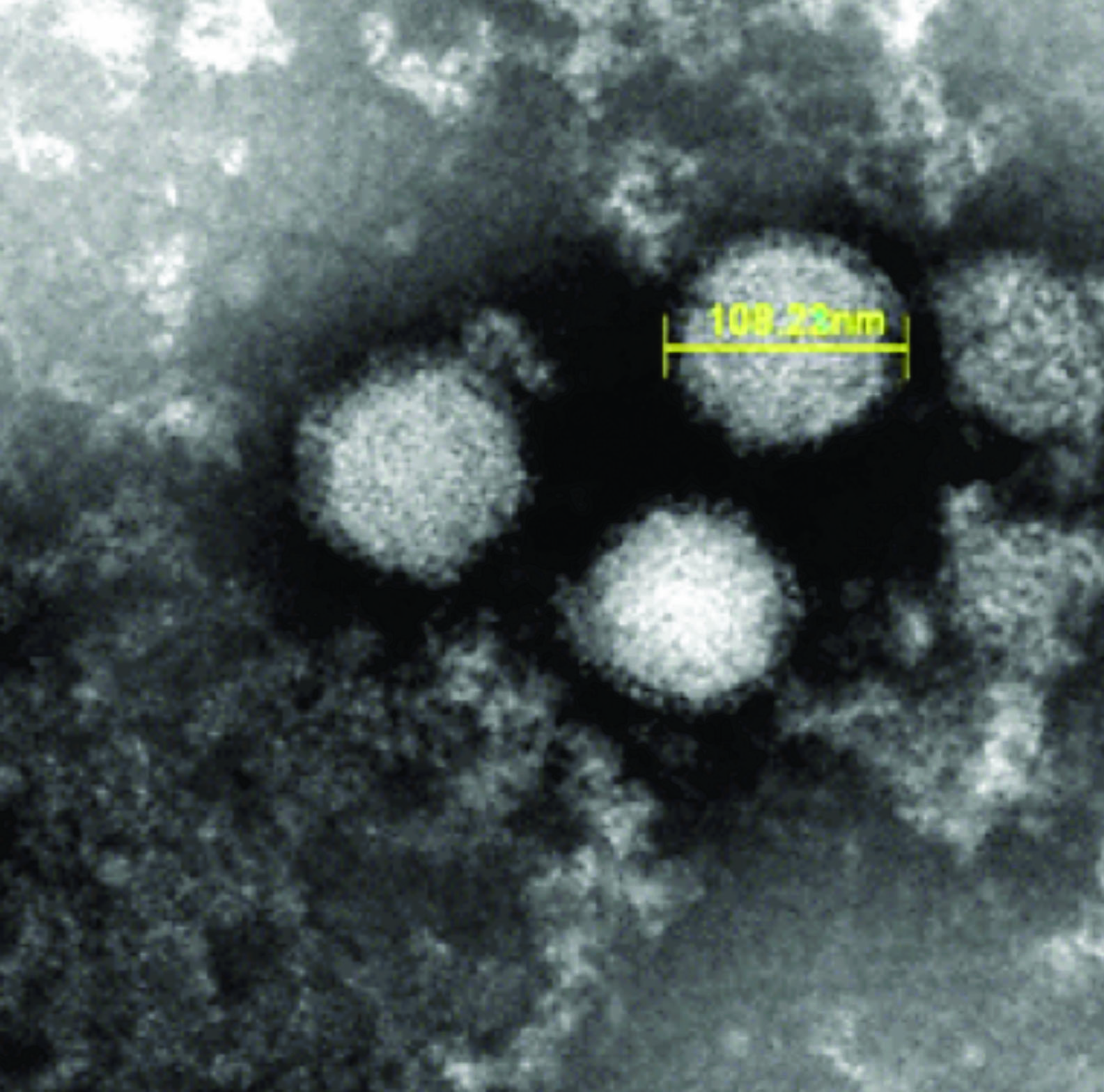
Image: A scanning electron micrograph (SEM) of severe fever with thrombocytopenia syndrome virus (Photo courtesy of the Japanese National Institute of Infectious Diseases).
Severe fever with thrombocytopenia syndrome (SFTS) is a tick-borne infectious disease with a high case fatality rate, and is caused by the SFTS virus (SFTSV) and the disease is endemic to China, South Korea, and Japan.
The viral ribonucleic acid (RNA) level in sera of patients with SFTS is known to be strongly associated with outcomes and therefore virological SFTS diagnosis with high sensitivity and specificity are required in disease endemic areas.
Scientists at the Japanese National Institute of Infectious Diseases (Tokyo, Japan) and their colleagues collected 63 serum samples from 55 acute phase patients suspected of SFTS in Japan. Viral gene detection by the quantitative reverse transcription polymerase chain reaction (qRT-PCR) and viral antibody detection by immunoglobulin G (IgG) enzyme-linked immunosorbent assay (ELISA) and/or indirect fluorescent antibody (IFA) were conducted to diagnose SFTS. From 55 patients, 34 of these were diagnosed as having SFTSV. Serum samples obtained from 18 healthy donors were used to establish the cut-off value of the IgG ELISA. Serum samples used for IgG ELISA were inactivated under the UV light in the biosafety cabinet for one hour.
The investigators generated novel monoclonal antibodies (MAbs) against the SFTSV nucleocapsid (N) protein and developed a sandwich antigen (Ag)-capture enzyme-linked immunosorbent assay (ELISA) for the detection of N protein of SFTSV using MAb and polyclonal antibody as capture and detection antibodies, respectively. The Ag-capture ELISAs were read using an optical density at 405 nm (OD405) was measured against a reference of 490 nm using a Model 680 Microplate Reader (Bio-Rad Laboratories Inc.; Hercules, CA, USA). The Ag-capture system was capable of detecting at least 350 to 1,220 50% Tissue Culture Infective Dose (TCID50)/100 μL/well from the culture supernatants of various SFTSV strains.
All 24 serum samples (100%) containing high copy numbers of viral RNA more than 105 copies/mL) showed a positive reaction in the Ag-capture ELISA, whereas 12 out of 15 serum samples (80%) containing low copy numbers of viral RNA (less than 105 copies/mL) showed a negative reaction in the Ag-capture ELISA. Among these Ag-capture ELISA- negative 12 samples, nine (75%) were positive for IgG antibodies against SFTSV. The authors conclude that the newly developed Ag-capture ELISA is useful for SFTS diagnosis in acute phase patients with high levels of viremia. The study was published on April 5, 2016, in the journal Public Library of Science Neglected Tropical Diseases.
The viral ribonucleic acid (RNA) level in sera of patients with SFTS is known to be strongly associated with outcomes and therefore virological SFTS diagnosis with high sensitivity and specificity are required in disease endemic areas.
Scientists at the Japanese National Institute of Infectious Diseases (Tokyo, Japan) and their colleagues collected 63 serum samples from 55 acute phase patients suspected of SFTS in Japan. Viral gene detection by the quantitative reverse transcription polymerase chain reaction (qRT-PCR) and viral antibody detection by immunoglobulin G (IgG) enzyme-linked immunosorbent assay (ELISA) and/or indirect fluorescent antibody (IFA) were conducted to diagnose SFTS. From 55 patients, 34 of these were diagnosed as having SFTSV. Serum samples obtained from 18 healthy donors were used to establish the cut-off value of the IgG ELISA. Serum samples used for IgG ELISA were inactivated under the UV light in the biosafety cabinet for one hour.
The investigators generated novel monoclonal antibodies (MAbs) against the SFTSV nucleocapsid (N) protein and developed a sandwich antigen (Ag)-capture enzyme-linked immunosorbent assay (ELISA) for the detection of N protein of SFTSV using MAb and polyclonal antibody as capture and detection antibodies, respectively. The Ag-capture ELISAs were read using an optical density at 405 nm (OD405) was measured against a reference of 490 nm using a Model 680 Microplate Reader (Bio-Rad Laboratories Inc.; Hercules, CA, USA). The Ag-capture system was capable of detecting at least 350 to 1,220 50% Tissue Culture Infective Dose (TCID50)/100 μL/well from the culture supernatants of various SFTSV strains.
All 24 serum samples (100%) containing high copy numbers of viral RNA more than 105 copies/mL) showed a positive reaction in the Ag-capture ELISA, whereas 12 out of 15 serum samples (80%) containing low copy numbers of viral RNA (less than 105 copies/mL) showed a negative reaction in the Ag-capture ELISA. Among these Ag-capture ELISA- negative 12 samples, nine (75%) were positive for IgG antibodies against SFTSV. The authors conclude that the newly developed Ag-capture ELISA is useful for SFTS diagnosis in acute phase patients with high levels of viremia. The study was published on April 5, 2016, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Japanese National Institute of Infectious Diseases
Bio-Rad Laboratories
Latest Microbiology News
- Study Finds Hidden Mpox Infections May Drive Ongoing Spread
- Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
- Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
- Rapid Antigen Biosensor Detects Active Tuberculosis in One Hour
- Label-Free Microscopy Method Enables Faster, Quantitative Detection of Malaria
- Oral–Gut Microbiome Signatures Identify Early Gastric Cancer
- Gut Microbiome Test Predicts Melanoma Recurrence After Surgery
- Rapid Blood-Culture Susceptibility Panel Expands Coverage for Gram-Negative Infections
- Antibiotic Resistance Genes Found in Newborns Within Hours of Birth
- Rapid Color Test Stratifies Virulent and Resistant Staph Strains
- mNGS CSF Test Identifies CNS Pathogens Missed by Standard Panels
- Syndromic Panel Enables Rapid Identification of Bloodstream Infections
- RNA-Based Workflow Identifies Active Skin Microbes for Dermatology Research
- Cost-Effective Sampling and Sequencing Workflow Identifies ICU Infection Hotspots
- New Bacterial Target Identified for Early Detection of Noma
- Genomic Analysis Links Emerging Streptococcal Strains to Specific Infections
Channels
Clinical Chemistry
view channel
Urine-Based Nanosensor Tracks Lung Cancer and Fibrosis Noninvasively
Lung cancer remains difficult to monitor for early progression and treatment resistance, while pulmonary fibrosis continues to pose major challenges for early diagnosis. Clinicians need repeatable, noninvasive... Read more
Blood-Based Alzheimer’s Test Gains CE Mark for Amyloid Pathology Detection
Alzheimer’s disease is the most common cause of dementia, yet confirmatory testing remains invasive and hard to access. Diagnosis currently takes an average of 3.5 years, and about 75% of people with dementia... Read moreMolecular Diagnostics
view channel
ctDNA Blood Test Could Help Guide Radiotherapy in Patients with Limited Metastases
Selecting the right therapy for patients whose solid tumors have begun to spread remains a major clinical challenge. Clinicians often count metastatic lesions on X-ray, computed tomography (CT), or magnetic... Read more
FDA-Approved MRD Blood Test Guides Adjuvant Bladder Cancer Therapy
Muscle-invasive bladder cancer (MIBC) carries a substantial risk of recurrence after radical cystectomy, and selecting which patients require adjuvant therapy remains challenging. Approximately 30,000... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreMicrobiology
view channel
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read morePathology
view channel
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read more
AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
Non–muscle invasive bladder cancer has highly variable outcomes, complicating surveillance and treatment planning. Risk assessment typically relies on stage, grade, and tumor size, leaving uncertainty... Read moreTechnology
view channel
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read more
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read moreIndustry
view channel
AI-Powered Multi-Functional Analyzer Wins German Innovation Award
Hematology services are increasingly delivered across distributed care settings, where limited staffing and complex workflows can extend turnaround times. Advanced morphology review still often depends... Read more