Innovative Nano-Biosensor Developed for Rapid Detection of Viruses
|
By LabMedica International staff writers Posted on 23 Mar 2016 |

Image: A novel nano-biosensor has been developed for lower-cost, rapid virus detection based on upconversion luminescence resonance energy transfer (LRET) technology and DNA oligo hybridization. Testing takes only 2–3 hours, about 10x faster traditional clinical methods (Photo courtesy of the Hong Kong Polytechnic University).
Scientists have invented a compact, easy-to-use biosensor for rapid, sensitive detection of flu and other viruses by utilizing the optical method of upconversion luminescence resonance energy transfer (LRET).
The new method, invented by researchers at the Hong Kong Polytechnic University (PolyU; Hong Kong), involves simple operational procedures that significantly reduce testing time from around 1–3 days to 2–3 hours, making it over 10 times quicker than traditional clinical methods. Furthermore, it is a low-cost test—about 80% lower than traditional tests, and the technology can be widely used to detect different types of viruses
RT-PCR is expensive and time-consuming while the sensitivity for ELISA is relatively low. Such limitations make these methods difficult for use in front-line and on-site virus detection. This challenge paved the way for the researchers to develop a new biosensor based on the luminescent technique.
The luminescent technique operates analogous to two matching pieces of magnet with attraction force. It involves the development of upconversion nanoparticles (UCNPs) conjugated with an oligo probe whose DNA base pairs are complementary with that of the gold nanoparticles (AuNPs) flu virus oligo. Given the complementarity, the two oligos undergo DNA-DNA hybridization. Upon being illuminated by a portable near-infrared laser pen, the UCNPs emit eye-visible green light while the AuNPs would absorb the green light. One can easily quantify the concentration of the targeted flu virus by measuring the decrease in green light intensity.
Initially, the researchers used upconversion LRET for ultrasensitive virus detection in liquid-phase system. The team then further improved the sensitivity by using a solid-phased nanoporous membrane system (NAAO). As NAAO membrane consists of many hollow channels, it allows more space for oligo hybridization to take place, increasing sensitivity by over 10 folds compared to the liquid-phase system, based on detection using inactivated virus samples.
The new biosensor does not require expensive instruments and sophisticated operational skills, and has sensitivity comparable to traditional clinical methods. In comparison to conventional downconversion luminescent technique, it causes low damage to genetic materials and does not induce background fluorescence. In addition, a complementary probe can be designed to target detection of any virus with known genetic sequence. In other words, the new method can be widely used for the detection of different types of viruses simply by modifying the UCNPs capture probe.
The team will continue to enhance the biosensor for virus detection, including increasing sensitivity and specificity, and developing a matrix for multiplex detection of multiple flu viruses on a single testing platform.
The related results have been recently published in the two nanomaterial research journals ACS Nano and Small.
Related Links:
The Hong Kong Polytechnic University
The new method, invented by researchers at the Hong Kong Polytechnic University (PolyU; Hong Kong), involves simple operational procedures that significantly reduce testing time from around 1–3 days to 2–3 hours, making it over 10 times quicker than traditional clinical methods. Furthermore, it is a low-cost test—about 80% lower than traditional tests, and the technology can be widely used to detect different types of viruses
RT-PCR is expensive and time-consuming while the sensitivity for ELISA is relatively low. Such limitations make these methods difficult for use in front-line and on-site virus detection. This challenge paved the way for the researchers to develop a new biosensor based on the luminescent technique.
The luminescent technique operates analogous to two matching pieces of magnet with attraction force. It involves the development of upconversion nanoparticles (UCNPs) conjugated with an oligo probe whose DNA base pairs are complementary with that of the gold nanoparticles (AuNPs) flu virus oligo. Given the complementarity, the two oligos undergo DNA-DNA hybridization. Upon being illuminated by a portable near-infrared laser pen, the UCNPs emit eye-visible green light while the AuNPs would absorb the green light. One can easily quantify the concentration of the targeted flu virus by measuring the decrease in green light intensity.
Initially, the researchers used upconversion LRET for ultrasensitive virus detection in liquid-phase system. The team then further improved the sensitivity by using a solid-phased nanoporous membrane system (NAAO). As NAAO membrane consists of many hollow channels, it allows more space for oligo hybridization to take place, increasing sensitivity by over 10 folds compared to the liquid-phase system, based on detection using inactivated virus samples.
The new biosensor does not require expensive instruments and sophisticated operational skills, and has sensitivity comparable to traditional clinical methods. In comparison to conventional downconversion luminescent technique, it causes low damage to genetic materials and does not induce background fluorescence. In addition, a complementary probe can be designed to target detection of any virus with known genetic sequence. In other words, the new method can be widely used for the detection of different types of viruses simply by modifying the UCNPs capture probe.
The team will continue to enhance the biosensor for virus detection, including increasing sensitivity and specificity, and developing a matrix for multiplex detection of multiple flu viruses on a single testing platform.
The related results have been recently published in the two nanomaterial research journals ACS Nano and Small.
Related Links:
The Hong Kong Polytechnic University
Latest Technology News
- Noninvasive Sputum Test Detects Early Lung Cancer
- New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
- Rapid Biosensor Detects Drug Sensitivity in Breast Tumors
- Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
- Online Tool Supports Family Screening for Inherited Cancer Risk
- Portable Breath Sensor Detects Pneumonia Biomarkers in Minutes
- New Electronic Pipette Enhances Workflows with Touchscreen Control
- AI Model Outperforms Clinicians in Rare Disease Detection
- AI-Driven Diagnostic Demonstrates High Accuracy in Detecting Periprosthetic Joint Infection
- Blood Test “Clocks” Predict Start of Alzheimer’s Symptoms
- AI-Powered Biomarker Predicts Liver Cancer Risk
Channels
Clinical Chemistry
view channel
Study Finds ApoB Testing More Effective Than LDL for Guiding Lipid Therapy
Routine blood tests that measure low-density lipoprotein (LDL), commonly known as “bad” cholesterol, are widely used to guide lipid-lowering therapy, but they do not always provide a complete picture of... Read more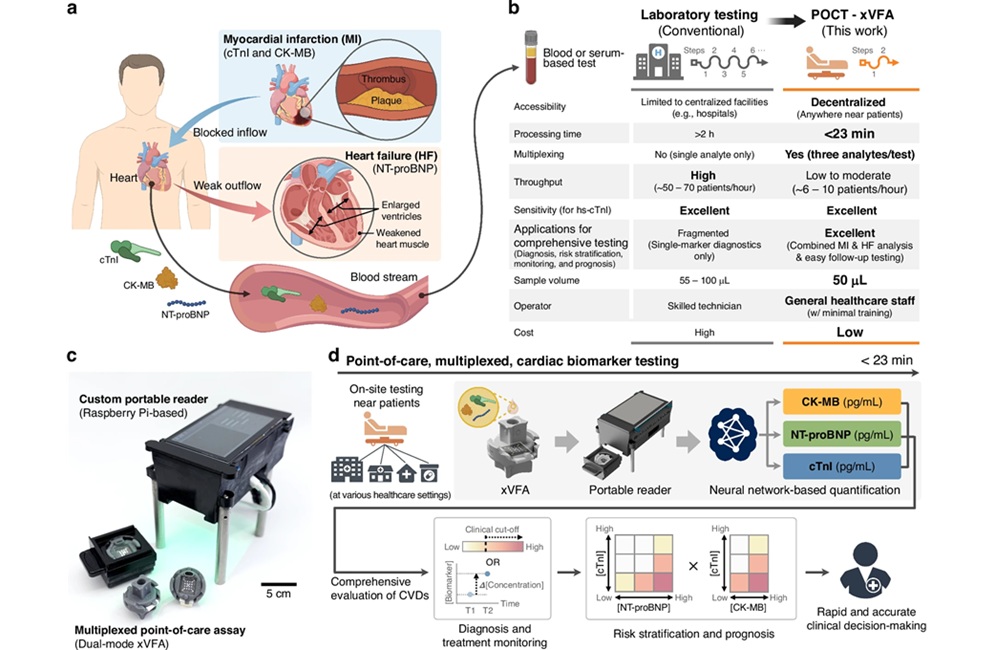
AI-Enabled POC Test Quantifies Multiple Cardiac Biomarkers
Cardiovascular diseases are a leading cause of death, responsible for nearly 20 million deaths each year. Timely triage of myocardial infarction and heart failure hinges on rapid cardiac biomarker measurement,... Read moreNext Generation Automated Analyzers Increase Throughput for Clinical Chemistry and Electrolyte Testing
Clinical laboratories continue to face staffing shortages, limited space, and growing test volumes that pressure chemistry and electrolyte workflows. Maintaining rapid turnaround times increasingly depends... Read moreMolecular Diagnostics
view channel
Urine-Based Assay Predicts Severe Dengue Risk Early
Dengue is among the most widespread mosquito-borne infections, yet early risk stratification can be difficult when symptoms overlap with other febrile illnesses. Only about 2% to 5% of patients develop... Read more
CE-Marked Blood Test Enables Monitoring of Neuroinflammation in Multiple Sclerosis
Multiple sclerosis affects more than 2.9 million people worldwide and requires ongoing evaluation of disease activity to guide care. Routine monitoring often depends on magnetic resonance imaging (MRI),... Read more
Ultrasensitive Assay Tracks Resistance Mutations MRD Monitoring
Ultrasensitive circulating tumor DNA assessment is being applied to predict and track response to immunotherapy and to detect relapse across multiple solid tumors. Monitoring resistance mutations such... Read more
Extracellular Vesicle RNA Biomarkers Enable Noninvasive IBD Diagnosis and Monitoring
Inflammatory bowel disease (IBD) is a chronic, relapsing gastrointestinal condition whose incidence is rising in industrialized and newly industrialized countries, with prevalence in early‑industrialized... Read moreHematology
view channel
Routine Blood Test Parameters Link Anemia to Cancer Risk and Mortality
Anemia detected in routine care can signal underlying pathology and is frequently encountered in adults. Because it is defined by hemoglobin levels below the normal range, it is often evaluated with red... Read more
Prognostic Tool Guides Personalized Treatment in Rare Blood Cancer
Chronic myelomonocytic leukemia (CMML) is a rare blood cancer in which acquired genetic mutations in bone marrow stem cells drive disease. Stem cell transplantation is the only curative option but carries... Read moreImmunology
view channel
Study Finds Influenza Often Undiagnosed in Winter Deaths
Seasonal influenza drives substantial excess mortality, yet its contribution is often obscured when infections go undiagnosed near the time of death. Many deaths occur outside hospitals or in older adults... Read moreCombined Screening Approach Identifies Early Leprosy Cases
Leprosy remains a significant public health concern, with more than 200,000 new cases reported globally each year and early disease often escaping routine laboratory detection. In its initial phase, bacterial... Read morePathology
view channel
Biomarker Predicts Immunotherapy Response and Prognosis in Colorectal Cancer
Colorectal cancer is common and often lethal, and therapeutic decision-making is complicated by heterogeneous tumor microenvironments. Immunotherapy benefits only a small subset of patients, around 5%,... Read moreAI Improves Completeness of Complex Cancer Pathology Reports
Oncology teams increasingly rely on pathology reports that integrate histopathology, immunohistochemistry, and rapidly expanding biomarker testing. As patients live longer and undergo repeated analyses... Read more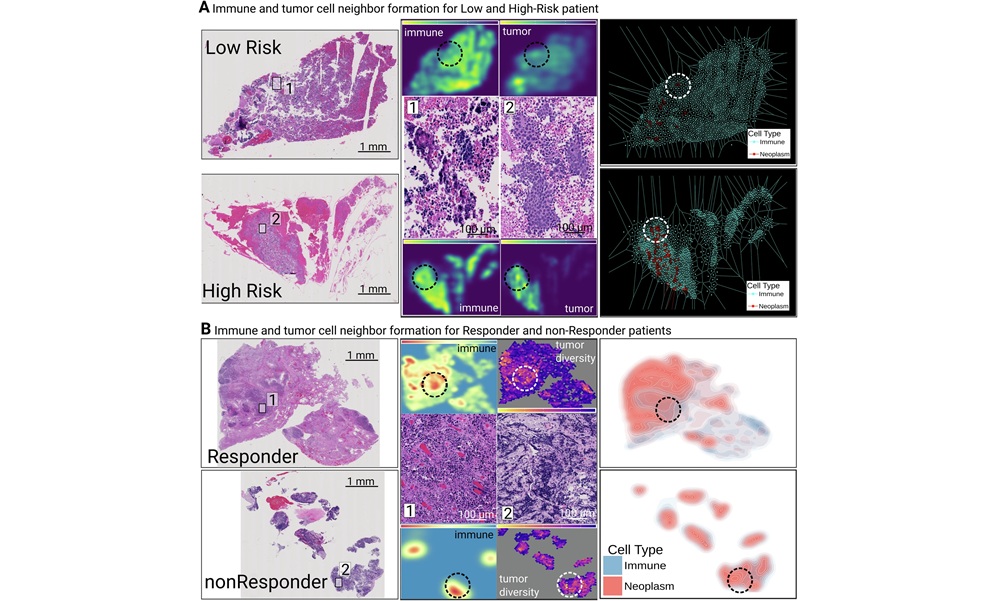
AI Tool Predicts Chemotherapy Response in Small Cell Lung Cancer
Small cell lung cancer often presents at an extensive stage and progresses rapidly, leaving little time to tailor first-line therapy. Clinicians currently lack biomarkers to guide which patients will benefit... Read more_image.png)
Tumor-Specific Biomarker Predicts Neoadjuvant Immunotherapy Response in Gastric Cancer
Gastric cancer is the fifth most common malignancy and the fourth leading cause of cancer mortality worldwide, with China bearing nearly half of the global burden. Only a subset of patients benefit from... Read moreTechnology
view channel
Noninvasive Sputum Test Detects Early Lung Cancer
Early detection remains critical for improving outcomes in lung cancer, yet clinicians increasingly encounter indeterminate pulmonary nodules found incidentally or through screening, complicating decision-making.... Read more
New AI Tool Enables Rapid Treatment Selection in Pediatric Leukemia
Children with T-cell acute lymphoblastic leukemia face an aggressive disease that remains difficult to treat. Although remission rates have improved, many survivors experience long-term effects from intensive... Read more
Breakthrough Mass Spectrometry Design Could Enable Ultra-Low Abundance Detection
Mass spectrometry is central to identifying and quantifying molecules in complex biological samples, but conventional instruments typically analyze ions sequentially, which can limit detection of rare species.... Read moreIndustry
view channel
Beckman Coulter Gains CE Mark for Rapid Assay Distinguishing Bacterial vs Viral Infections
Clinicians often struggle to distinguish bacterial from viral infections at first presentation because symptoms overlap and definitive culture or molecular results can take hours or days.... Read more