Rapid Urine Test Detects Early Kidney Injury
|
By LabMedica International staff writers Posted on 05 May 2014 |
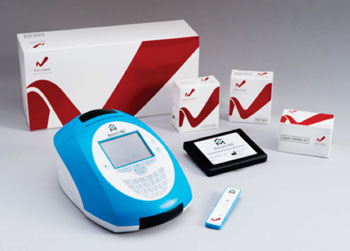
Image: The NephroCheck Test cartridge and Astute140 Meter (Photo courtesy of Astute Medical).
A new test utilizes fluorescence immunoassay technology to detect and measure two biomarkers that reflect the risk of developing acute kidney injury (AKI).
The NephroCheck Test measures tissue inhibitor of metalloproteinase 2 (TIMP-2) and insulin-like growth factor binding protein 7(IGFBP-7) in human urine. Both are involved in G1 cell cycle arrest during the very early phases of cellular stress, and are robust measures of risk for AKI manifesting within the following 12-24 hours. TIMP-2 is an inhibitor of the matrix metalloproteinases (MMPs) that directly suppress the proliferation of endothelial cells. IGFBP-7 is involved in the regulation of tissue availability of insulin and IGFs, and stimulates cell adhesion implicated in epithelial cell-cycle arrest.
For the test, a sample of a patient’s urine is mixed with reagents onto a single-use cartridge inserted into the proprietary Astute140 Meter. The meter employs a sandwich immunoassay technique along with fluorescence detection technology to quantitatively measure the biomarker concentrations, combining them into a single numerical test result. Multiple levels of controls (internal, external, and electronic) are used to ensure reliable and accurate results in about 20 minutes. The NephroCheck Test and Astute140 Meter are products of Astute Medical (San Diego, CA, USA), and have received the European Community CE marking of approval.
“We believe the NephroCheck Test will be able to help physicians address one of the most costly, deadly, and confounding conditions affecting intensive care hospital patients,” said Chris Hibberd, CEO of Astute Medical. “With increasing constraints on healthcare spending, innovative tools such as the NephroCheck Test will be essential to ensuring that the right resources get to the right patients at the right time.”
AKI is a significant global health hazard that strikes up to seven percent of hospitalized patients. AKI often occurs in patients that are already suffering from myriad issues such as sepsis, trauma, surgery, or being treated with nephrotoxic drugs, further complicating diagnosis, while also increasing mortality rates.
Related Links:
Astute Medical
The NephroCheck Test measures tissue inhibitor of metalloproteinase 2 (TIMP-2) and insulin-like growth factor binding protein 7(IGFBP-7) in human urine. Both are involved in G1 cell cycle arrest during the very early phases of cellular stress, and are robust measures of risk for AKI manifesting within the following 12-24 hours. TIMP-2 is an inhibitor of the matrix metalloproteinases (MMPs) that directly suppress the proliferation of endothelial cells. IGFBP-7 is involved in the regulation of tissue availability of insulin and IGFs, and stimulates cell adhesion implicated in epithelial cell-cycle arrest.
For the test, a sample of a patient’s urine is mixed with reagents onto a single-use cartridge inserted into the proprietary Astute140 Meter. The meter employs a sandwich immunoassay technique along with fluorescence detection technology to quantitatively measure the biomarker concentrations, combining them into a single numerical test result. Multiple levels of controls (internal, external, and electronic) are used to ensure reliable and accurate results in about 20 minutes. The NephroCheck Test and Astute140 Meter are products of Astute Medical (San Diego, CA, USA), and have received the European Community CE marking of approval.
“We believe the NephroCheck Test will be able to help physicians address one of the most costly, deadly, and confounding conditions affecting intensive care hospital patients,” said Chris Hibberd, CEO of Astute Medical. “With increasing constraints on healthcare spending, innovative tools such as the NephroCheck Test will be essential to ensuring that the right resources get to the right patients at the right time.”
AKI is a significant global health hazard that strikes up to seven percent of hospitalized patients. AKI often occurs in patients that are already suffering from myriad issues such as sepsis, trauma, surgery, or being treated with nephrotoxic drugs, further complicating diagnosis, while also increasing mortality rates.
Related Links:
Astute Medical
Latest Clinical Chem. News
- Urine-Based Nanosensor Tracks Lung Cancer and Fibrosis Noninvasively
- FDA-Cleared Assay Enables Comprehensive Automated Testosterone Testing
- CE-Marked Blood Biomarker Test Advances Automated Alzheimer’s Diagnostics
- Blood-Based Alzheimer’s Test Gains CE Mark for Amyloid Pathology Detection
- Noninvasive Urine Test May Support Earlier Diagnosis of Psychiatric Disorders
- At-Home Blood and Cognitive Tests Support Dementia Risk Stratification
- Ultrasensitive Test Detects Key Biomarker of Frontotemporal Dementia Subtype
- Routine Blood Tests Years Before Pregnancy Could Identify Preeclampsia Risk
- Blood Test Detects Testicular Cancer Missed by Standard Markers
- Routine Blood Tests Identify Biomarkers Linked to PTSD
- Proteomic Data Underscore Need for Age-Specific Pediatric Reference Ranges
- Routine Blood Count Ratio Linked to Future Alzheimer’s and Dementia Risk
- Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
- Rapid Biosensor Detects Pancreatic Cancer Biomarker for Early Detection
- Urine-Based Multi-Cancer Screening Test Receives FDA Breakthrough Device Designation
- Blood Test Predicts Alzheimer Disease Risk Before Imaging Changes and Symptoms
Channels
Molecular Diagnostics
view channel
ctDNA Blood Test Could Help Guide Radiotherapy in Patients with Limited Metastases
Selecting the right therapy for patients whose solid tumors have begun to spread remains a major clinical challenge. Clinicians often count metastatic lesions on X-ray, computed tomography (CT), or magnetic... Read more
FDA-Approved MRD Blood Test Guides Adjuvant Bladder Cancer Therapy
Muscle-invasive bladder cancer (MIBC) carries a substantial risk of recurrence after radical cystectomy, and selecting which patients require adjuvant therapy remains challenging. Approximately 30,000... Read moreHematology
view channel
Stem Cell Biomarkers May Guide Precision Treatment in Acute Myeloid Leukemia
Acute myeloid leukemia (AML) is an aggressive blood cancer that most often affects older adults and still carries a poor prognosis despite therapeutic advances. Venetoclax-based regimens have improved... Read more
Advanced CBC-Derived Indices Integrated into Hematology Platforms
Diatron, a STRATEC brand, has introduced six advanced hematological indices on its Aquila, Aquarius 3, and Abacus 5 hematology analyzers. The new Research Use Only (RUO) indices include Neutrophil-to-Lymphocyte... Read moreImmunology
view channel
Routine TB Screening Test May Reveal Immune Aging and Mortality Risk
Immune aging is associated with weaker responses to vaccination, greater risks of infection, and higher levels of inflammation. Leveraging routinely ordered laboratory tests to quantify that responsiveness... Read more
Biomarkers and Molecular Testing Advance Precision Allergy Care
Allergic diseases often present with similar symptoms but can be driven by distinct biological mechanisms, making standardized care inefficient for many patients. Historically, individuals with pollen... Read moreMicrobiology
view channel
Study Finds Hidden Mpox Infections May Drive Ongoing Spread
Mpox continues to circulate despite vaccination, and many cases show no known link to a symptomatic partner. The role of people without symptoms has remained uncertain, limiting clarity on how transmission persists.... Read more
Large-Scale Genomic Surveillance Tracks Resistant Bacteria Across European Hospitals
Antimicrobial resistance (AMR) poses a growing threat to patient safety, with carbapenem-resistant Enterobacterales causing difficult-to-treat infections and leaving clinicians with limited therapeutic options.... Read more
Molecular Urine and Stool Tests Do Not Improve Early TB Treatment in Hospitalized HIV Patients
Tuberculosis is the leading cause of death among people living with HIV, and diagnosis in hospital settings remains difficult. Symptoms are often non-specific, disease can be extrapulmonary, and many patients... Read morePathology
view channel
Rapid AI Tool Predicts Cancer Spatial Gene Expression from Pathology Images
Gene expression profiling can inform tumor biology and treatment selection, but spatial assays remain costly and time-consuming. Results can take weeks and cost thousands of dollars, limiting large-scale... Read more
AI Pathology Test Receives FDA Breakthrough for Bladder Cancer Risk Stratification
Non–muscle invasive bladder cancer has highly variable outcomes, complicating surveillance and treatment planning. Risk assessment typically relies on stage, grade, and tumor size, leaving uncertainty... Read moreTechnology
view channel
Point-of-Care Testing Enhances Health Literacy and Self-Management in Chronic Disease
Limited access to general practitioners and pathology services can delay diagnosis and monitoring for people in regional and remote communities. Rapid, on-the-spot testing can shorten turnaround times... Read more
Fully Automated Sample-to-Insight Workflow Advances Latent TB Testing
Latent tuberculosis remains a substantial testing workload for clinical laboratories as screening programs expand. Despite this growth, only about 40% of testing has shifted from traditional skin tests... Read moreIndustry
view channel
AI-Powered Multi-Functional Analyzer Wins German Innovation Award
Hematology services are increasingly delivered across distributed care settings, where limited staffing and complex workflows can extend turnaround times. Advanced morphology review still often depends... Read more