Liquid Biopsy Detects Breast Cancer Residual Disease
By LabMedica International staff writers
Posted on 02 Jan 2020
The concept of circulating DNA or tumor cells to identify patients with residual cancer present after early-stage, putatively curative treatment has made rapid strides across multiple tumor types since some of the first promising tests in colorectal cancer.Posted on 02 Jan 2020
Triple-negative breast cancers, unlike some other breast cancer types, carry significant risk of recurrence even when diagnosed at early stages. In addition, they lack the growing slate of targeted treatment options available to other molecular subsets and because of this, standard of care is limited to chemotherapy, radiation, and surgery.
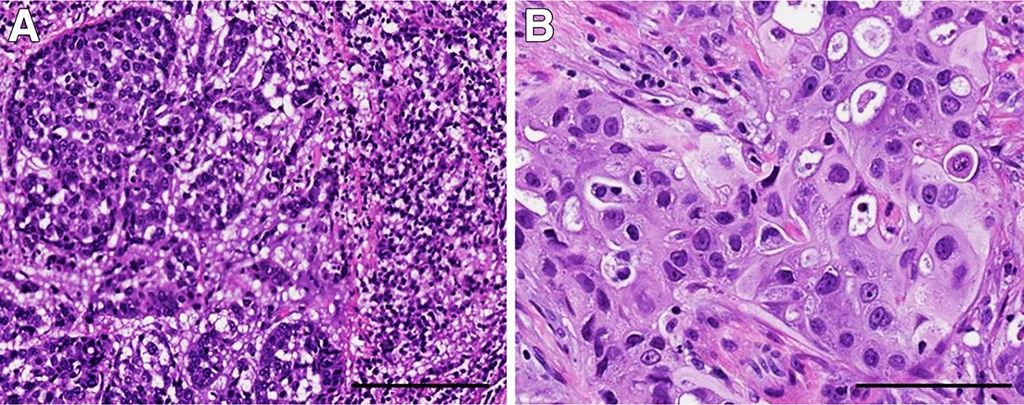
Image: Morphologic variants of triple-negative breast cancer (TNBC) with different genetic alterations. A: TNBC with basal-like histologic features containing a prominent stromal lymphocytic infiltrate; this tumor had MYC amplification. B: TNBC with apocrine differentiation and a PI3KCA mutation. The tumor cells have abundant eosinophilic cytoplasm, round nuclei, and prominent nucleoli (Photo courtesy of Geisel School of Medicine).
Scientists from the Indiana University School of Medicine (Indianapolis, IN, USA) and their colleagues analyzed retrospective plasma samples that had been collected from patients enrolled in the Phase II BRE12-158 clinical trial, which studied genomically directed therapy versus physician’s choice of treatment after preoperative chemotherapy in patients with high-risk triple-negative breast cancer.
The trial enrolled 196 women in total, 142 of whom had circulating tumor DNA (ctDNA) sequencing performed using the FoundationOne Liquid Test (Foundation Medicine, Cambridge, MA, USA) and enough clinical follow-up to study. Testing identified mutated ctDNA in 90 of the patients, about 60 %, with TP53 being the most commonly mutated gene, followed by others that are commonly associated with breast cancer.
At 17.2 months of follow up, the patients in whom ctDNA had been detected had significantly inferior distant disease free survival (DDFS) compared to those who didn't. The group showing circulating mutations survived without distant recurrences 32.5 months on average, while patients without ctDNA had not reached a median. At 24 months, the DDFS rate was 56% for ctDNA-positive patients, compared with 81% in ctDNA-negative patients. By combining ctDNA and circulating tumor cell detection boosted this even further. Patients who were double positive (having both ctDNA and circulating tumor cells present) had a two-year DDFS of just 52% compared to 89% for double negatives.
Milan Radovich, PhD, an associate professor and first author of the study said, “With neoadjuvant chemotherapy about one third of triple-negative patients achieve a state of pathologic complete response, in which there is no evidence of their tumor once surgeons go in to remove it. This subgroup has much better outcomes than the two thirds who still have residual disease after neoadjuvant chemo.” The study was presented at the San Antonio Breast Cancer Symposium held December 10 - 14, 2019, in San Antonio, TX, USA.
Related Links:
Indiana University School of Medicine
Foundation Medicine














