Noninvasive Test Monitors Bladder Cancer Recurrence
By LabMedica International staff writers
Posted on 19 Feb 2015
The results of a blinded, multicenter clinical study that used noninvasive technology to detect bladder cancer recurrence in patients with a history of the disease have been announced. Posted on 19 Feb 2015
The blinded clinical study was conducted in nine medical centers, where urine samples from 217 subjects with a history of bladder cancer were tested. The study population included 121 healthy subjects and 96 patients currently suffering from the disease.
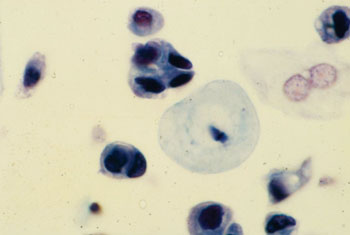
Image: Urothelial carcinoma cells of high malignant potential in voided urine sample (Photo courtesy of Michael Gordon, PhD).
The CellDetect test (Micromedic Technologies Ltd.; Tel Aviv, Israel) successfully identified cancerous cells in urine samples, with reported sensitivity of 84.4% and specificity of 82.7% for the study's primary endpoint. The results of the CellDetect urine test were compared with results from biopsy or cystoscopy, in cases where biopsies were not performed. The results also indicated that the negative predictive value (NPV) was 98.5%. In addition to its high sensitivity for advanced stage tumors and high-grade malignancy, the test was also found to exhibit high sensitivity for early stage tumors and low-grade malignancies which are difficult to identify using other noninvasive tests currently available on the market.
The secondary endpoint showed that the sensitivity of other noninvasive comparator tests, urine cytology, BTA stat (Polymedco; Redmond, WA, USA) and NMP22 BladderCheck (Alere; Waltham, MA, USA) was 50.0%, 68.8% and 17.4%, respectively. These findings indicate that the method is adequately sensitive for the purpose of accurate and early detection of the recurrence of the disease.
Ofer Yossepowitch. MD, Head of the Uro-Oncology Service at Rabin Medical Center (Petach Tikva, Israel), said, “The study results are encouraging. The accuracy of this novel assay appears to be superior over any available noninvasive test, suggesting a potential to supplant some or all of the cystoscopies required for bladder cancer surveillance. This is indeed great news for patients with history of bladder cancer, which may change their management.”
Related Links:
Micromedic Technologies Ltd.
Polymedco
Alere














