Multiplex Antibody Assay Could Transform Hepatitis B Immunity Testing
Posted on 02 Jan 2026
Hepatitis B remains a major global health challenge, yet immunity testing has historically been constrained by cost, operational complexity, and single-analyte approaches. Now, a multiplex antibody assay enables simultaneous detection of antibodies to hepatitis B, HIV-1, HIV-2, and hepatitis C from a single sample, reducing turnaround time, conserving patient samples, and supporting evidence-based clinical decision-making.
Pictor’s (Carlsbad, CA, USA) ViraScreen-Core multiplex antibody assay, built for both serum and saliva, offers unmatched flexibility for labs while addressing broader infectious disease screening needs. CDC's Advisory Committee on Immunization Practices (ACIP) has recommended post-vaccination antibody testing for hepatitis B, resulting in laboratories facing new demands to assess immunity more effectively.
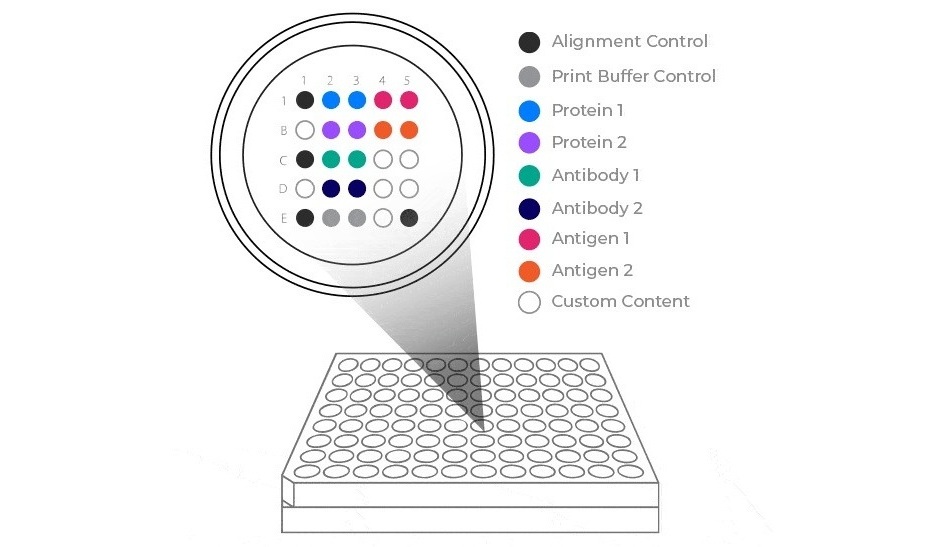
ViraScreen-Core, an established test already in use by CLIA-certified laboratories, offers a ready solution to support this evolving public health guidance. Pictor has completed paired serum and saliva studies demonstrating high concordance, supporting saliva as a reliable testing matrix. This capability may help reduce barriers to testing, particularly in pediatric, community health, and decentralized care settings where blood draws can be challenging.
"Antibody testing gives laboratories flexibility as public health guidance evolves," said Dr Jamie Platt, CEO of Pictor. "By offering a multiplex assay that works with both serum and saliva, we aim to help labs move toward evidence-based immunity assessment without adding operational complexity."
"Pictor's ViraScreen-Core assay represents a valuable addition to our clinical laboratory," said Dr Andrew Brown, Chief Commercial Officer at Sirona Dx. "Compatibility with saliva samples expands the environments in which testing can be conducted while the multiplex format supports streamlined, efficient workflows for infectious disease screening."
Related Links:
Pictor