Alzheimer's Disease Meta-Analysis Identifies New Risk Genes
By LabMedica International staff writers
Posted on 11 Mar 2019
The risk for late-onset Alzheimer’s disease (LOAD), the most prevalent dementia, is partially driven by genetics. Scientists have uncovered five new late-onset Alzheimer's disease risk genes in a genetic meta-analysis. They likewise confirmed 20 other genes previously linked to late-onset Alzheimer's risk.Posted on 11 Mar 2019
A very large team of scientists led by those at the University of Miami Miller School of Medicine (Miami, FL, USA) combined 46 different datasets to yield an overall set of 35,274 clinical and autopsy-documented Alzheimer's cases and 59,163 controls to interrogate. Through the first two stages of their multi-stage analysis, they uncovered 21 loci reaching genome-wide significance, 18 of which had previously been identified. After the third stage of their investigation and an overall meta-analysis, they identified 13 novel loci associated with risk of late-onset Alzheimer's.
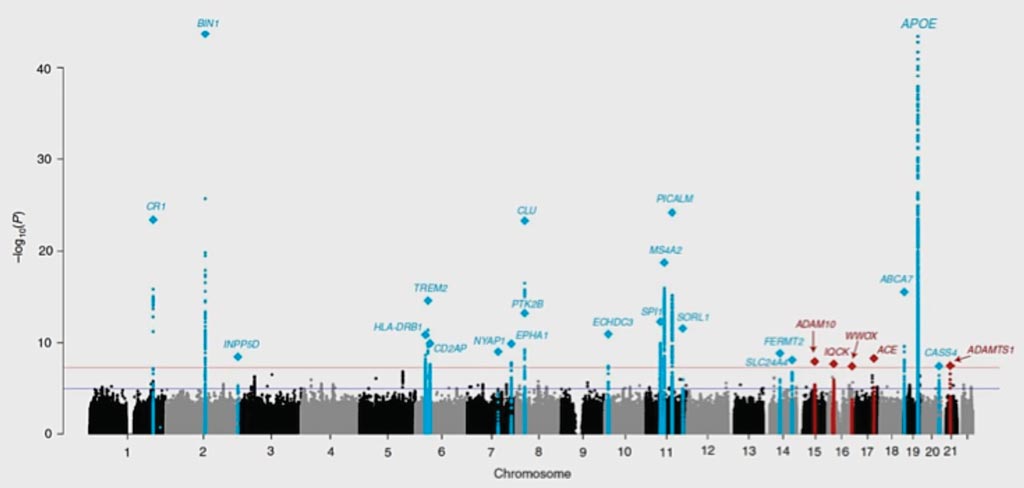
Image: Newly identified (red) and known (blue) genes linked to Alzheimer’s disease spike in this table plotting results from genome-wide association analysis of 94,437 individuals with late onset Alzheimer’s (Photo courtesy of Brian W. Kunkle, MD, PhD and colleagues).
They identified the genes through a genome-wide association meta-analysis using data from more than 94,000 people, which also enabled them to tie certain biological processes like immune response, lipid metabolism, tau binding proteins, and amyloid precursor protein metabolism to disease. This, they noted, suggests that variants affecting amyloid precursor protein (APP) and beta amyloid (Aβ) processing play roles in both early- and late-onset Alzheimer's. Amyloid plaques of Aβ protein and neurofibrillary tangles of tau protein are disease hallmarks.
The team combined together various line of evidence, such as gene annotation, expression quantitative trait loci (eQTL) analyses, gene expression analysis, and pathway analyses, they prioritized candidate genes at the novel signals they found, implicating the IQCK, ACE, ADAM10, ADAMTS1, and WWOX genes in Alzheimer's risk. ADAM10, for instance, encodes an alpha secretase that is active in the brain and is part of the non-amyloidogenic pathway of APP. WWOZ, which is a high-density-lipoprotein cholesterol and triglyceride-associated gene, is highly expressed in astrocytes and neurons, and binds tau, where it may regulate tau hyper-phosphorylation, the formation of neurofibrillaries, and aggregation of Aβ.
The team noted that pathway analysis showing that tau is involved in late-onset Alzheimer's supports recent evidence that tau may play an early pathological role in the disease and confirms that therapies targeting tangle formation or degradation could potentially affect late-onset disease. Their analysis of risk genes and disease-linked pathways also indicated an enrichment of rare variants in late-onset Alzheimer's disease risk and suggested there might be additional rare variants to find if larger samples are studied. The study was published on February 28, 2019, in the journal Nature Genetics.
Related Links:
University of Miami Miller School of Medicine




.jpg)