LAMP Accurately Screens for Malaria in Pregnancy
By LabMedica International staff writers
Posted on 24 Jul 2018
Pregnant women are especially susceptible to Plasmodium infections and have the risk of developing severe disease and birth complications. These might include maternal anemia, intra-uterine growth retardation, infant low birth weight, prematurity, miscarriage and stillbirth.Posted on 24 Jul 2018
Pregnant women frequently show low-density Plasmodium infections that require more sensitive methods for accurate diagnosis and early treatment of malaria. This is particularly relevant in low-malaria transmission areas, where intermittent preventive treatment is not recommended.
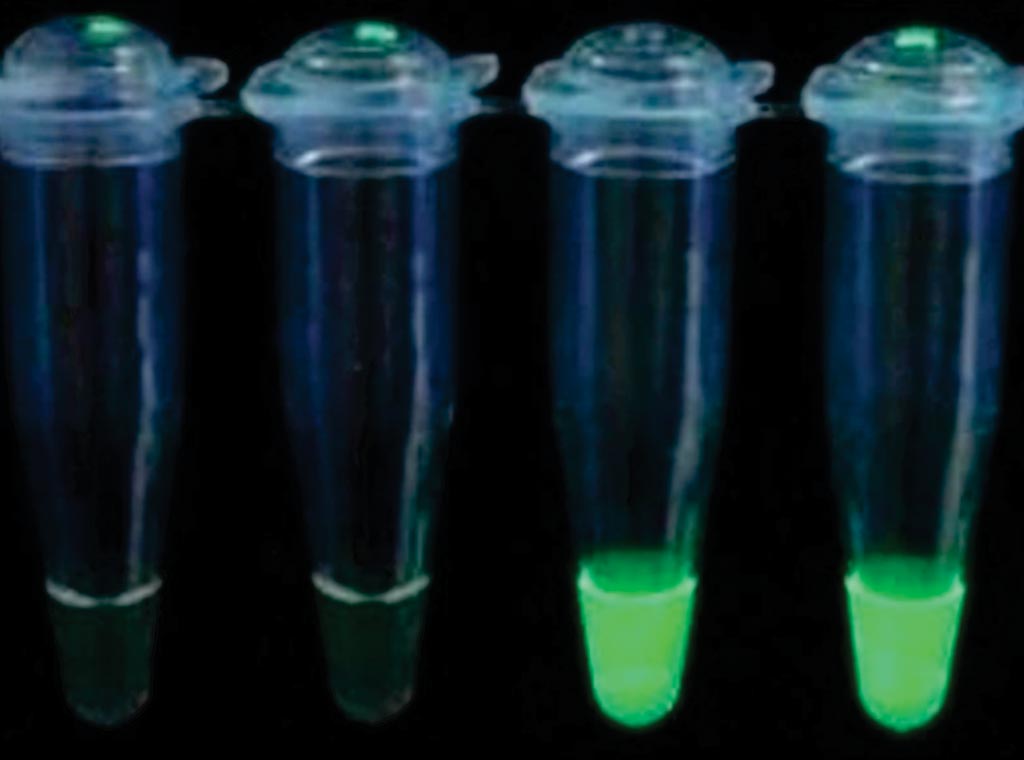
Image: The Loop Mediated Isothermal Amplification (LAMP) test for the detection of malaria under ultra-violet light, positives two right tubes (Photo courtesy of HUMAN Diagnostics).
Scientists at the Universidad de Antioquia (Medellín, Colombia) and their international colleagues carried out a nested prospective study that used data and samples from a larger cross-sectional project conducted from May 2016 to January 2017 in three Colombian endemic areas. A total of 531 peripheral and placental samples were collected from pregnant women self-presenting at local hospitals for antenatal care visits, at delivery or seeking medical care for suspected malaria.
Blood samples were analyzed for Plasmodium parasites by light microscopy (LM), rapid diagnostic test (RDT) and loop mediated isothermal amplification (LAMP). The team used RDT SD Bioline Malaria antigen Pf/Pv as it can distinguish between P. falciparum, P. vivax and mixed infections. The commercially available Loopamp MALARIA Pan/Pf detection kit consists of reaction tubes with ready-made vacuum-dried reagents for the detection of P. falciparum (Pf-LAMP) or Plasmodium spp. (Pan-LAMP). DNA was extracted from half blood-spot filters and processed for a universal polymerase chain reaction (PCR) followed by nested species-specific PCR (nPCR) to detect the 18S ribosomal gene of P. falciparum, P. vivax and P. malariae.
The team reported that in peripheral samples, LAMP showed an improved sensitivity of 100% when compared with LM 79.5% and RDT 76.9%, particularly in afebrile women, for which LAMP sensitivity was, two-times higher than LM and RDT. Overall agreement among LAMP and nPCR was high. In placental blood, LAMP evidenced a four-fold improvement in sensitivity (88.9%) when compared with LM and RDT (22.2%), being the only method, together with nPCR, able to detect placental infections in peripheral blood.
The authors concluded that LAMP is a simple, rapid and accurate molecular tool for detecting gestational and placental malaria, being able to overcome the limited sensitivity of LM and RDT. These findings could guide maternal health programs in low-transmission settings to integrate LAMP in their surveillance systems for the active detection of low-density infections and asymptomatic malaria cases. The study was published on July 13, 2018, in the Malaria Journal.
Related Links:
Universidad de Antioquia














