Common Metablolic Enzyme Could Predict Response to Cancer Immunotherapy
Posted on 06 Apr 2026
Immune checkpoint inhibitors can generate durable tumor control, but only about 20% of patients benefit and clinicians still lack robust tools to predict response. Tumors also exploit metabolic programs and immune-evasion pathways that blunt therapeutic efficacy. Identifying actionable biomarkers for programmed cell death protein 1 (PD-1) or programmed death-ligand 1 (PD‑L1) therapies remains a priority for oncology and pathology services. A new study shows that a common metabolic enzyme may help forecast response and guide combination strategies.
At Rutgers Cancer Institute of New Jersey, investigators identified the metabolic enzyme phosphoglycerate dehydrogenase (PHGDH) as a potential predictor of immunotherapy benefit, with findings published in Cell Reports Medicine on March 28, 2026. PHGDH, which supports serine biosynthesis, was shown to increase PD‑L1 expression, enabling tumor immune evasion. The work also indicates that PHGDH levels are frequently elevated in cancers such as colorectal and breast tumors.
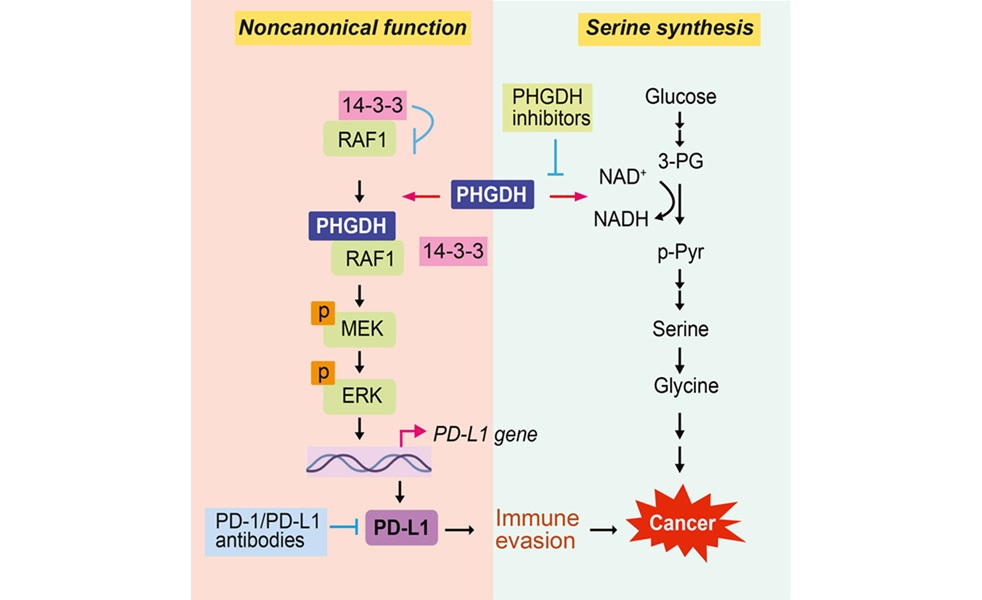
Mechanistically, the team demonstrated that PHGDH’s effect on PD‑L1 is independent of its catalytic role in serine production. Genetically altered, catalytically inactive versions of PHGDH still drove PD‑L1 upregulation, underscoring a noncanonical function for this enzyme. Because PD‑L1 mediates immune escape, its linkage to PHGDH suggests a direct molecular bridge between cancer metabolism and checkpoint signaling.
Preclinical testing combined an experimental PHGDH inhibitor with an immune checkpoint agent. In mouse studies conducted over 60 days, approximately half of animals receiving the two‑drug regimen survived without observed side effects. In contrast, about 20% survived with either monotherapy, and none survived without treatment.
The researchers also analyzed clinical datasets from patients previously treated with PD‑1/PD‑L1 therapies and found significantly better responses in tumors with high PHGDH expression. If confirmed in a large prospective trial, measuring PHGDH could help oncologists and laboratory teams identify patients most likely to benefit from checkpoint inhibitors. The study was conducted at Rutgers Cancer Institute of New Jersey, together with RWJBarnabas Health.
“The effect was substantial compared with single drug treatment,” said Zhaohui Feng, senior author and professor at Rutgers Cancer Institute. "Those patients who have high PHGDH are more sensitive to anti-PD-1 therapy. This could be a potential biomarker to predict the efficacy of checkpoint inhibitor therapy."
Related Links
Rutgers Cancer Institute
RWJBarnabas Health