Screening Tool Simplifies Serological Diagnosis of Neurocysticercosis
By LabMedica International staff writers
Posted on 24 Jul 2014
The availability of a rapid serological diagnosis that targets stage-specific antibodies for human cysticercosis is considered very helpful in control programs for estimating the sero-prevalence or burden of the disease in susceptible population groups.Posted on 24 Jul 2014
Cysticercosis is a tissue infection caused by hatched oncospheres, a larval form of the pork tapeworm Taenia solium which parasitize the intestinal wall and circulate through the bloodstream, invade the nervous system and form cysts in the brain, leading to neurocysticercosis.
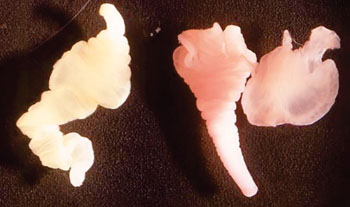
Image: Taenia solium cysticerci, which represent the larval or intermediate, immature developmental stages of this pork tapeworm (Photo courtesy of Dr. George R. Healy).
Medical parasitologists at the Leiden University Medical Center (The Netherlands) and their American colleagues developed a field-friendly screening tool to simplify serological diagnosis of neurocysticercosis. A total of 63 banked serum samples from patients with confirmed neurocysticercosis were used to validate the assay, and a total of 170 serum samples collected in regions where transmission of cysticercosis does not occur, were used to assess specificity. These samples consisted of a panel of 78 serum samples assembled from healthy residents of the USA and a panel of 92 serum samples from blood bank donors in the Netherlands.
The assay utilizes novel nano-sized up-converting phosphor (UCP) reporter particles in combination with a portable lightweight analyzer and detects antibodies in serum samples reactive with bacterial-expressed recombinant (r) T24H, a marker for detecting neurocysticercosis cases. Three sequential flow steps allow enrichment of antibodies on the Test (T) line and consecutive binding of protein-A coated UCP reporter particles. Antibody binding was determined by measuring 550-nm emission after excitation of the UCP label with a 980 nm infrared (IR) diode. Lateral flow (LF) strips were scanned using a multistrip scanner, modified Packard Fluorocount microplate reader (PerkinElmer; Waltham, MA, USA) and a customized ESEQuant lateral flow reader (LFR) (QIAGEN Lake Constance; Stockach, Germany).
The clinical sensitivity and specificity of the assay to detect cases of human neurocysticercosis with two or more viable brain cysts were 96% and 98%, respectively, using a sample set comprised of sera from 63 confirmed cases and 170 healthy parasite-naïve non-endemic controls. The evaluation of the new test with a set of clinical samples indicated excellent clinical parameters comparable to an enzyme-linked immunosorbent assay (ELISA). Application of new nano-sized UCP reporter label and a lightweight strip analyzer, both from easily accessible sources, demonstrated equivalent performance compared to previous studies. The study was published on July 3, 2014, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Leiden University Medical Center
PerkinElmer
QIAGEN














