High-Throughput Microfluidic Nanoimmunoassay Detects Anti-SARS-CoV-2 Antibodies in Serum or Ultra Low-Volume Blood Samples
By LabMedica International staff writers
Posted on 11 May 2021
A team of researchers has developed a high-throughput microfluidic nanoimmunoassay (NIA) that can detect anti-SARS-CoV-2 antibodies in serum or ultra low-volume blood samples.Posted on 11 May 2021
The NIA developed by researchers at Icahn School of Medicine at Mount Sinai (New York, NY, USA) can detect anti-SARS-CoV-2 IgG antibodies in 1,024 samples per device. To enable decentralized blood sample collection, the method can detect antibodies in a small drop of blood obtainable by finger pricking, and the blood can be collected and shipped with a simple, low-cost blood glucose test strip. The method achieved a specificity of 100% and a sensitivity of 98% based on the analysis of 289 human serum samples.
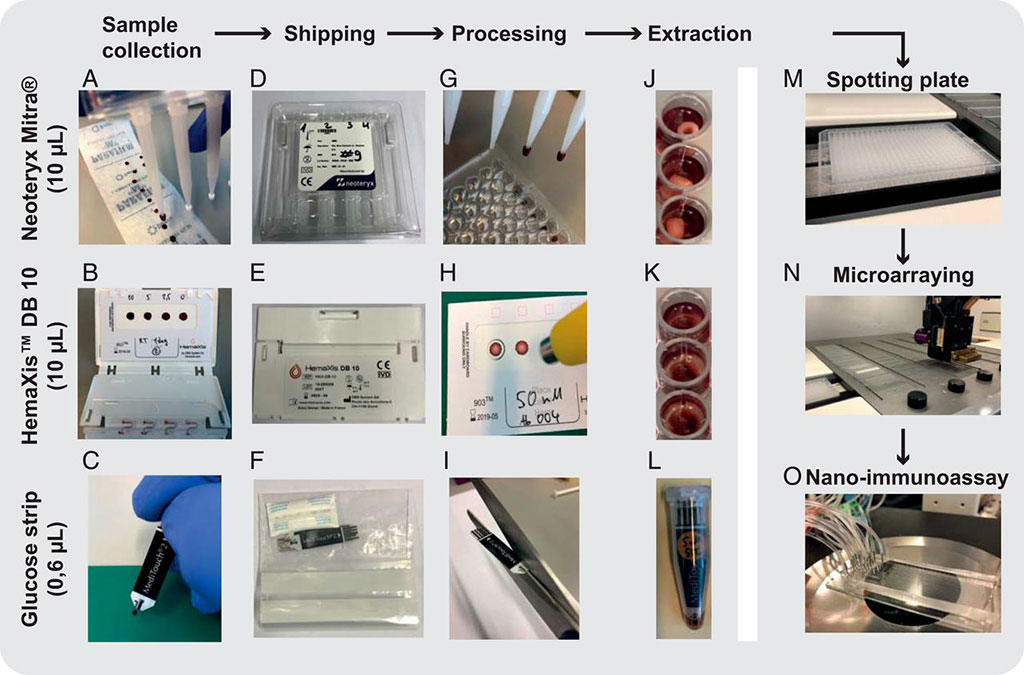
Image: Ultralow-volume whole blood sampling and processing (Photo courtesy of PNAS)
As the majority of people infected with SARS-CoV-2 have no or only mild symptoms, many cases aren’t captured by direct testing. However, it is important to establish the true spread of the virus by identifying how many people have been exposed. Detection of anti-SARS-CoV-2-specific antibodies in blood samples can help us understand how the pandemic is evolving over time. Novel technologies are needed to facilitate large-scale detection of SARS-CoV-2 specific antibodies in human blood samples. The novel technologies should be capable of high throughput, low reagent consumption, and low cost per test; achieve high sensitivity and specificity; and be compatible with ultra low-volume whole blood samples in the low or even submicroliter range that can be obtained via a simple finger prick. Such technologies are essential to support seroprevalence studies and vaccine clinical trials, and to monitor quality and duration of immunity.
To eliminate the need for venipuncture, the researchers developed low-cost, ultra low-volume whole blood sampling methods based on two commercial devices and repurposed a blood glucose test strip. The glucose test strip permits the collection, shipment, and analysis of 0.6 μL of whole blood easily obtainable from a simple finger prick. The NIA platform achieves high throughput, high sensitivity, and specificity based on the analysis of 289 human serum samples, and negligible reagent consumption. The researchers further demonstrated the possibility to combine NIA with decentralized and simple approaches to blood sample collection. They expect the technology to be applicable to current and future SARS-CoV-2 related serological studies and to protein biomarker analysis in general.
Related Links:
Icahn School of Medicine at Mount Sinai














