Beckman Coulter SARS-CoV-2 IgM Antibody Test Receives FDA Emergency Use Authorization
By LabMedica International staff writers
Posted on 12 Oct 2020
Beckman Coulter Diagnostics (Brea, CA, USA) has received Emergency Use Authorization (EUA) from the US Food & Drug Administration (FDA) for its Access SARS-CoV-2 Immunoglobulin M (IgM) assay.Posted on 12 Oct 2020
The assay detects antibodies that recognize the receptor binding domain (RBD) of the spike protein which the SARS-CoV-2 virus uses to enter the human host cells. The assay, which demonstrates 99.9% specificity and 98.3% sensitivity, is part of a suite of diagnostic solutions being developed by Beckman in response to the ongoing COVID-19 pandemic to guide physicians and patients in their healthcare decision making.
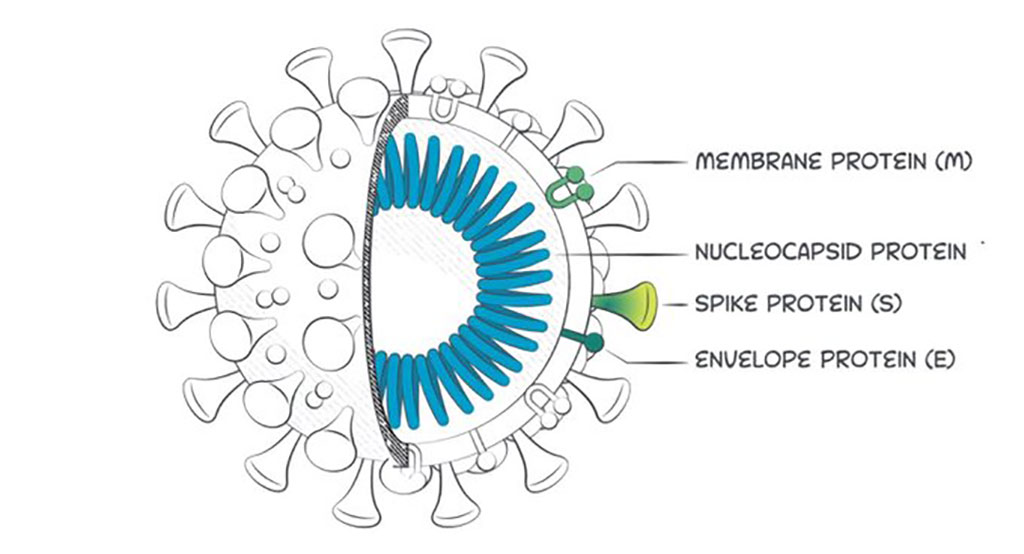
Image: Beckman Coulter SARS-CoV-2 IgM Antibody Test Receives FDA Emergency Use Authorization (Photo courtesy of Beckman Coulter)
Beckman’s suite of COVID-19 testing solutions includes the Access SARS-CoV-2 IgM assay and the Access SARS CoV-2 IgG assay, which received EUA in June. All of Beckman’s assays to address COVID-19 can be performed in automated or high-throughput immunoassay formats, as well as Beckman Coulter's Access 2 analyzer, a compact, table-top analyzer enabling high-quality serology testing to be carried out in small hospitals and clinics. Additionally, all of the assays seamlessly integrate into laboratory workflows making it easy to add these tests to routine blood tests performed during inpatient and wellness testing.
The company recently received FDA EUA for its interleukin 6 (IL-6) assay, which can be used to assist in identifying severe inflammatory response in patients with confirmed COVID 19 illness to aid in determining the risk of intubation with mechanical ventilation, in conjunction with clinical findings and the results of other laboratory testing. Beckman is also currently developing a SARS CoV-2 antigen assay as well as a quantitative IgG assay anticipated to be launched later this year.
"Since March, the Beckman Coulter team has worked around the clock to develop a suite of assays that play a critical role in the ongoing global fight against COVID-19," said Julie Sawyer Montgomery, president of Beckman Coulter. "As a science-driven company, we continue in our commitment to deliver rigorously validated diagnostics of the highest quality that provide meaningful information, so doctors and patients alike can trust the results for urgent, care decisions."














