BioFire Diagnostics’ PCR-based SARS-CoV-2 Test Receives FDA Emergency Use Authorization
By LabMedica International staff writers
Posted on 08 Oct 2020
BioMérieux’s (Marcy-l'Étoile, France) subsidiary, BioFire Diagnostics, has received Emergency Use Authorization by the US Food and Drug Administration for the BioFire Respiratory Panel 2.1-EZ which uses a syndromic approach to accurately detect and identify a wide range of pathogens, including SARS-CoV-2.Posted on 08 Oct 2020
The BioFire RP2.1-EZ Panel is designed to run on the CLIA-waived BioFire FilmArray 2.0 EZ Configuration System and identifies a menu of 19 respiratory targets in one multiplex PCR test, with results in about 45 minutes. Because a large number of pathogens cause respiratory infections, tests that only identify SARS-CoV-2 or influenza run the risk of missing the real culprit. With the ability to identify 15 viral and 4 bacterial respiratory pathogens, BioFire’s easy-to-use syndromic test requires just one nasopharyngeal swab and two minutes of hands-on time. The multiplex PCR BioFire RP2.1-EZ Panel provides a rapid, accurate, and comprehensive alternative to targeted testing and detects a greater number of pathogens no matter the season or the time of year.
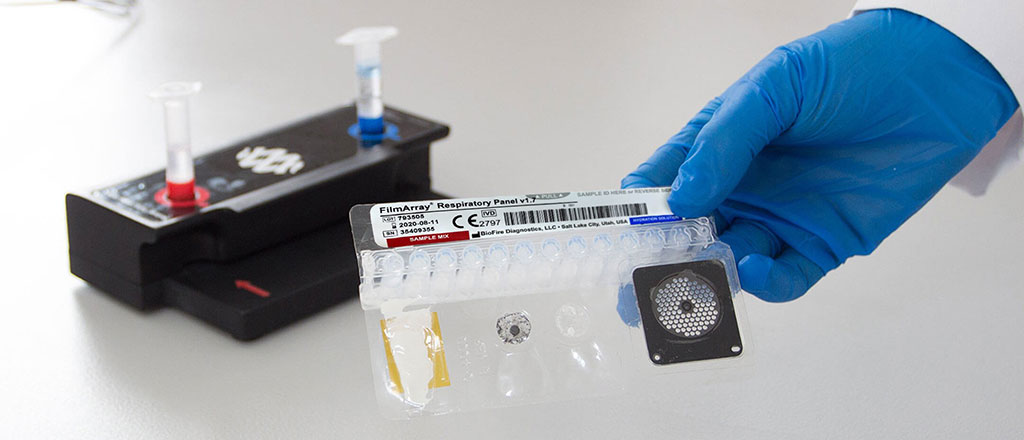
Image: BioFire Respiratory Panel 2.1-EZ (Photo courtesy of BioMérieux)
Given the shift toward value-based care, BioFire’s syndromic testing can help reduce untimely and expensive send-out tests. The BioFire RP2.1-EZ Panel (EUA) can help reduce overall workload for urgent care clinics, helping streamline workflow, maximize throughput, decrease costs, and increase patient satisfaction.
Related Links:
BioMérieux




.jpg)









