LAMP Test Rapidly Detects Sexually Transmitted Chlamydia
|
By LabMedica International staff writers Posted on 10 Jan 2019 |
Image: A High Pure PCR template preparation kit (Photo courtesy of Roche Diagnostics).
Chlamydia trachomatis is the leading cause of sexually transmitted diseases (STDs) in females and males in both developed and developing countries, with more than 110 million cases annually. C. trachomatis resists antibiotic treatment and is a cofactor in HIV transmission and human cervical cancer.
For nucleic acid amplification tests, recently loop-mediated isothermal amplification (LAMP) has presented an attractive alternative to standard methods like polymerase chain reaction (PCR) due to its low price, ease of use, rapid results, and lack of requirement for an expensive thermal cycler and specialized kits for DNA extraction and purification.
Scientists at the Chulalongkorn University (Bangkok, Thailand) and their associates have developed a rapid, inexpensive, easy-to-interpret, sensitive and specific point-of-care (POC) C. trachomatis detection system, using a LAMP assay for target C. trachomatis DNA amplification, followed by gold nanoparticle probe (AuNP) for colorimetric C. trachomatis specific readout.
The team collected endocervical swab samples were randomly selected from a prospective study cohort of sexually transmitted disease (STD) prevalence in symptomatic and healthy (which may include non-symptomatic patients) Thai women aged 15 to 54 years in Bangkok and nearby areas. The samples were collected by clinicians during 2011 and 2012 from qualified volunteers. The sample size of 130 (96 symptomatic and 34 healthy) was computed based on a standard statistical formula.
A set of six primers was designed for LAMP to target eight distinct regions on the C. trachomatis ompA gene. To determine the optimal LAMP assay condition (incubation temperature and incubation time), the LAMP reaction was incubated at 59 °C to 65 °C for 30 to 60 minutes (without loop primers) and 10 to 35 minutes (with loop primers). A 25 μL LAMP reaction comprised DNA template, High Pure PCR Template Preparation Kit. Nanogold particles of 10 nm were appended to the probe to create the complementary AuNP-DNA probe specific for the C. trachomatis LAMP product.
Of the 130 clinical samples, the LAMP-AuNP sensitivity was 96% (23/24) and specificity 99% (105/106). This result is higher than the LAMP-GE. The percentages of assay accuracy for PCR-GE and LAMP-AuNP were equal (98%), whereas for LAMP-GE it was 96%. The AuNPs allow simple visualization of results and improve the LAMP specificity and sensitivity. The percentages of assay accuracy for the independent replicate detection were 100% for PCR-GE, 99% for LAMP-AuNP, and 98% for LAMP-GE. Verification from the UV-vis spectra supported the convenient colorimetric reading of the LAMP-AuNP assay.
The authors concluded that the C. trachomatis LAMP-AuNP detection is appropriate for local and resource-constrained settings. The assay directly uses clinical samples, obtains results in 32 to 47 minutes and the result is readable by the naked eye. The study was published on December 20, 2018, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Chulalongkorn University
For nucleic acid amplification tests, recently loop-mediated isothermal amplification (LAMP) has presented an attractive alternative to standard methods like polymerase chain reaction (PCR) due to its low price, ease of use, rapid results, and lack of requirement for an expensive thermal cycler and specialized kits for DNA extraction and purification.
Scientists at the Chulalongkorn University (Bangkok, Thailand) and their associates have developed a rapid, inexpensive, easy-to-interpret, sensitive and specific point-of-care (POC) C. trachomatis detection system, using a LAMP assay for target C. trachomatis DNA amplification, followed by gold nanoparticle probe (AuNP) for colorimetric C. trachomatis specific readout.
The team collected endocervical swab samples were randomly selected from a prospective study cohort of sexually transmitted disease (STD) prevalence in symptomatic and healthy (which may include non-symptomatic patients) Thai women aged 15 to 54 years in Bangkok and nearby areas. The samples were collected by clinicians during 2011 and 2012 from qualified volunteers. The sample size of 130 (96 symptomatic and 34 healthy) was computed based on a standard statistical formula.
A set of six primers was designed for LAMP to target eight distinct regions on the C. trachomatis ompA gene. To determine the optimal LAMP assay condition (incubation temperature and incubation time), the LAMP reaction was incubated at 59 °C to 65 °C for 30 to 60 minutes (without loop primers) and 10 to 35 minutes (with loop primers). A 25 μL LAMP reaction comprised DNA template, High Pure PCR Template Preparation Kit. Nanogold particles of 10 nm were appended to the probe to create the complementary AuNP-DNA probe specific for the C. trachomatis LAMP product.
Of the 130 clinical samples, the LAMP-AuNP sensitivity was 96% (23/24) and specificity 99% (105/106). This result is higher than the LAMP-GE. The percentages of assay accuracy for PCR-GE and LAMP-AuNP were equal (98%), whereas for LAMP-GE it was 96%. The AuNPs allow simple visualization of results and improve the LAMP specificity and sensitivity. The percentages of assay accuracy for the independent replicate detection were 100% for PCR-GE, 99% for LAMP-AuNP, and 98% for LAMP-GE. Verification from the UV-vis spectra supported the convenient colorimetric reading of the LAMP-AuNP assay.
The authors concluded that the C. trachomatis LAMP-AuNP detection is appropriate for local and resource-constrained settings. The assay directly uses clinical samples, obtains results in 32 to 47 minutes and the result is readable by the naked eye. The study was published on December 20, 2018, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Chulalongkorn University
Latest Microbiology News
- Enhanced Rapid Syndromic Molecular Diagnostic Solution Detects Broad Range of Infectious Diseases
- Clinical Decision Support Software a Game-Changer in Antimicrobial Resistance Battle
- New CE-Marked Hepatitis Assays to Help Diagnose Infections Earlier
- 1 Hour, Direct-From-Blood Multiplex PCR Test Identifies 95% of Sepsis-Causing Pathogens
- Mouth Bacteria Test Could Predict Colon Cancer Progression
- Unique Metabolic Signature Could Enable Sepsis Diagnosis within One Hour of Blood Collection
- Groundbreaking Diagnostic Platform Provides AST Results With Unprecedented Speed
- Simple Blood Test Combined With Personalized Risk Model Improves Sepsis Diagnosis
- Blood Analysis Predicts Sepsis and Organ Failure in Children
- TB Blood Test Could Detect Millions of Silent Spreaders
- New Blood Test Cuts Diagnosis Time for Nontuberculous Mycobacteria Infections from Months to Hours
- New Tuberculosis Test to Expand Testing Access in Low- and Middle-Income Countries
- Rapid Test Diagnoses Tropical Disease within Hours for Faster Antibiotics Treatment
- Rapid Molecular Testing Enables Faster, More Targeted Antibiotic Treatment for Pneumonia
- Rapid AST Platform Provides Targeted Therapeutic Results Days Faster Than Current Standard of Care
- New Analysis Method Detects Pathogens in Blood Faster and More Accurately by Melting DNA
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more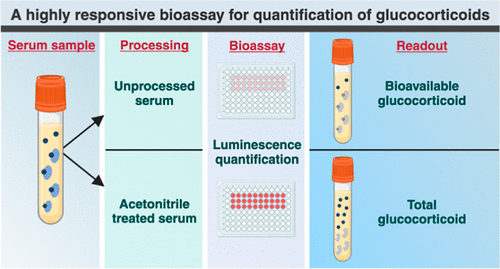
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreMolecular Diagnostics
view channel
Simple Blood Test Could Enable First Quantitative Assessments for Future Cerebrovascular Disease
Cerebral small vessel disease is a common cause of stroke and cognitive decline, particularly in the elderly. Presently, assessing the risk for cerebral vascular diseases involves using a mix of diagnostic... Read more
New Genetic Testing Procedure Combined With Ultrasound Detects High Cardiovascular Risk
A key interest area in cardiovascular research today is the impact of clonal hematopoiesis on cardiovascular diseases. Clonal hematopoiesis results from mutations in hematopoietic stem cells and may lead... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more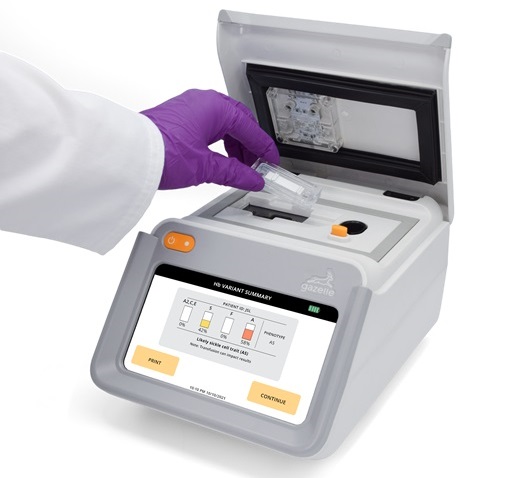
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more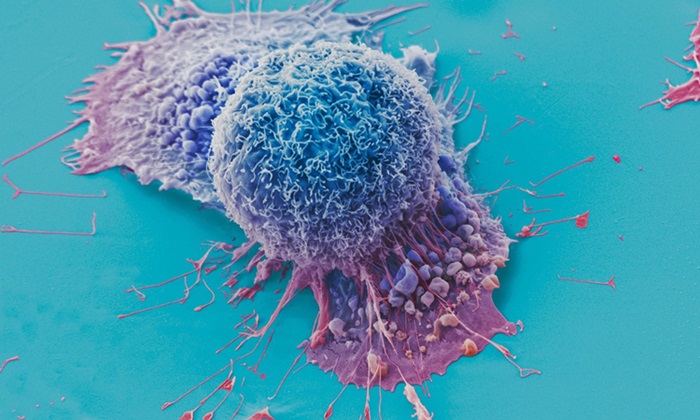
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read morePathology
view channel.jpg)
Use of DICOM Images for Pathology Diagnostics Marks Significant Step towards Standardization
Digital pathology is rapidly becoming a key aspect of modern healthcare, transforming the practice of pathology as laboratories worldwide adopt this advanced technology. Digital pathology systems allow... Read more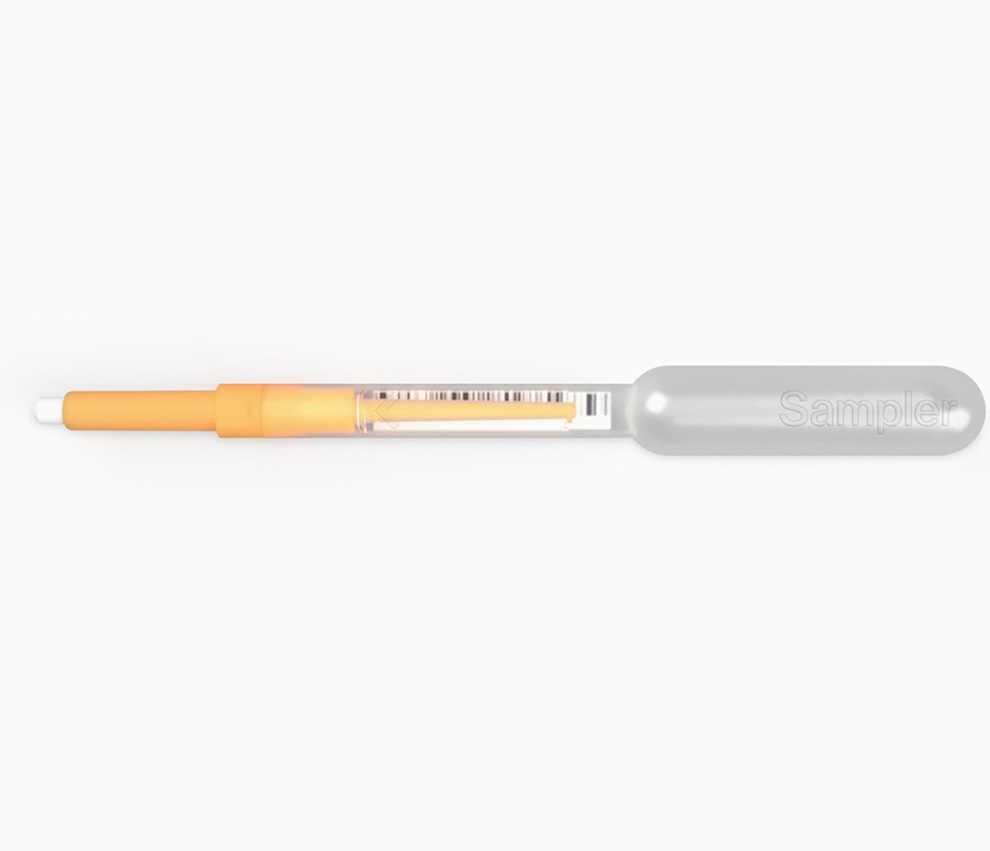
First of Its Kind Universal Tool to Revolutionize Sample Collection for Diagnostic Tests
The COVID pandemic has dramatically reshaped the perception of diagnostics. Post the pandemic, a groundbreaking device that combines sample collection and processing into a single, easy-to-use disposable... Read moreAI-Powered Digital Imaging System to Revolutionize Cancer Diagnosis
The process of biopsy is important for confirming the presence of cancer. In the conventional histopathology technique, tissue is excised, sliced, stained, mounted on slides, and examined under a microscope... Read more
New Mycobacterium Tuberculosis Panel to Support Real-Time Surveillance and Combat Antimicrobial Resistance
Tuberculosis (TB), the leading cause of death from an infectious disease globally, is a contagious bacterial infection that primarily spreads through the coughing of patients with active pulmonary TB.... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more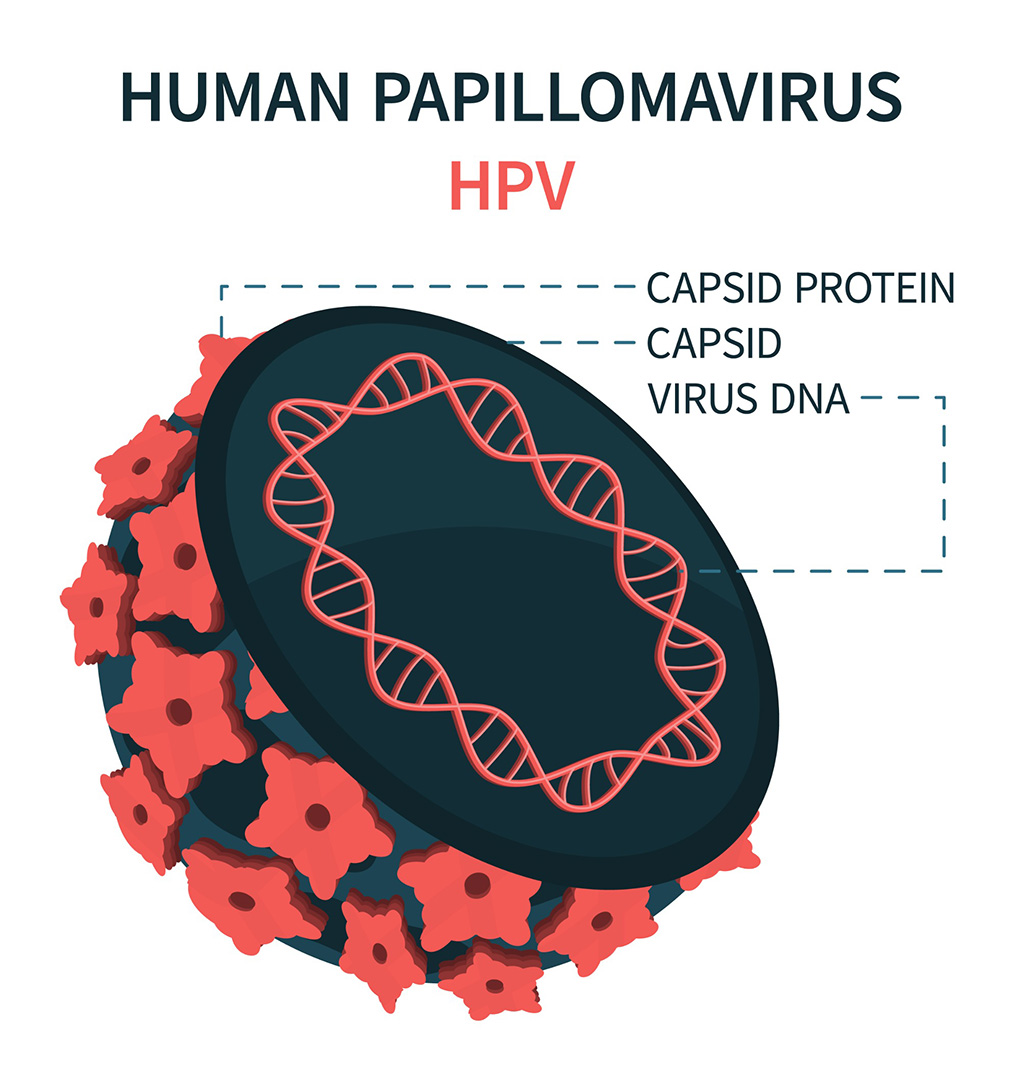
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more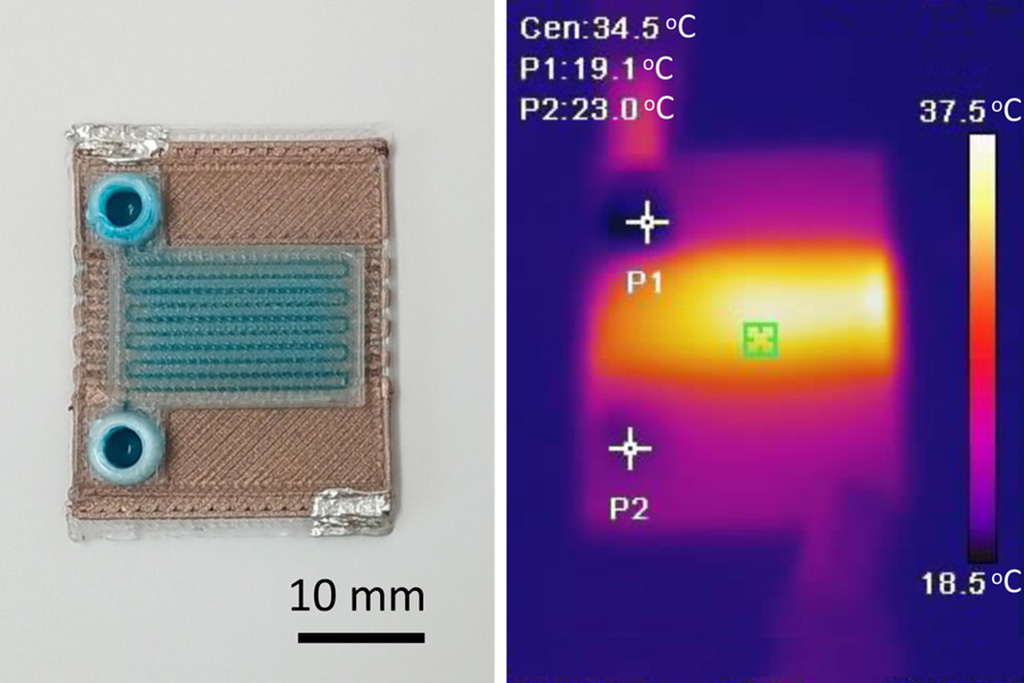
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel_1.jpg)
Thermo Fisher and Bio-Techne Enter Into Strategic Distribution Agreement for Europe
Thermo Fisher Scientific (Waltham, MA USA) has entered into a strategic distribution agreement with Bio-Techne Corporation (Minneapolis, MN, USA), resulting in a significant collaboration between two industry... Read more
ECCMID Congress Name Changes to ESCMID Global
Over the last few years, the European Society of Clinical Microbiology and Infectious Diseases (ESCMID, Basel, Switzerland) has evolved remarkably. The society is now stronger and broader than ever before... Read more