Multiplex Nucleic Acid Suspension Bead Array Detects Filoviruses
|
By LabMedica International staff writers Posted on 28 Apr 2015 |
Image: The SuperScript III One-Step reverse transcriptase polymerase chain reaction (RT-PCR) kit with Platinum Taq (Photo courtesy of Invitrogen).
Filoviruses (FVs) are ribonucleic acid (RNA) viruses that belong to the family Filoviridae, which includes zoonotic pathogens of the three genera Ebolavirus (EV), Marburgvirus (MV), and Cuevavirus (CV).
Multiplex suspension bead array systems that allow fast and reliable detection of reverse transcriptase polymerase chain reaction (RT-PCR) amplified filovirus genomes and enabled subtyping of Ebolavirus species and Marburgvirus strains has been described.
Scientists at the Public Health Agency of Sweden (Solna, Sweden) and their colleagues set up a multiplex suspension bead array system, by designing two different RT-PCRs that could amplify all known EV and MV species, respectively, based on whole-genome sequences of FVs, including Reston ebolavirus (RESTV), and Bundibugyo ebolavirus (BDBV). The RT-PCRs were performed using a SuperScript III One-Step RT-PCR kit with Platinum Taq (Invitrogen, Carlsbad CA, USA).
The RT-PCR amplicons were also sequenced to confirm specificity. In parallel, different sets of magnetic Bio-Plex Pro COOH beads (Bio-Rad Laboratories, Hercules, CA, USA) were coupled with oligonucleotides specific for each EV species and MV strain. The suspension bead arrays were also tested on human sera spiked with Lassa virus, Dengue virus serotypes 1 to 4, Crimean-Congo hemorrhagic fever virus, Rift Valley fever virus, and 10 FV-negative human sera. The results did not reveal any cross-reactivity which demonstrated the utility of the microsphere-based assay as a rapid and accurate diagnostic tool for confirming suspected FV infections.
The EV system can distinguish between three EV species and the MV system can distinguish between four MV strains. Based on the sequence information from the current Ebola outbreak in West Africa, the EBOV probe described in the study will also detect that particular strain. The multiplex suspension bead array system developed can easily be adapted to emerging new strains by including additional beads. The study was published in the April 2015 issue of the Journal of Clinical Microbiology.
Related Links:
Public Health Agency of Sweden
Invitrogen
Bio-Rad Laboratories
Multiplex suspension bead array systems that allow fast and reliable detection of reverse transcriptase polymerase chain reaction (RT-PCR) amplified filovirus genomes and enabled subtyping of Ebolavirus species and Marburgvirus strains has been described.
Scientists at the Public Health Agency of Sweden (Solna, Sweden) and their colleagues set up a multiplex suspension bead array system, by designing two different RT-PCRs that could amplify all known EV and MV species, respectively, based on whole-genome sequences of FVs, including Reston ebolavirus (RESTV), and Bundibugyo ebolavirus (BDBV). The RT-PCRs were performed using a SuperScript III One-Step RT-PCR kit with Platinum Taq (Invitrogen, Carlsbad CA, USA).
The RT-PCR amplicons were also sequenced to confirm specificity. In parallel, different sets of magnetic Bio-Plex Pro COOH beads (Bio-Rad Laboratories, Hercules, CA, USA) were coupled with oligonucleotides specific for each EV species and MV strain. The suspension bead arrays were also tested on human sera spiked with Lassa virus, Dengue virus serotypes 1 to 4, Crimean-Congo hemorrhagic fever virus, Rift Valley fever virus, and 10 FV-negative human sera. The results did not reveal any cross-reactivity which demonstrated the utility of the microsphere-based assay as a rapid and accurate diagnostic tool for confirming suspected FV infections.
The EV system can distinguish between three EV species and the MV system can distinguish between four MV strains. Based on the sequence information from the current Ebola outbreak in West Africa, the EBOV probe described in the study will also detect that particular strain. The multiplex suspension bead array system developed can easily be adapted to emerging new strains by including additional beads. The study was published in the April 2015 issue of the Journal of Clinical Microbiology.
Related Links:
Public Health Agency of Sweden
Invitrogen
Bio-Rad Laboratories
Latest Molecular Diagnostics News
- Novel Biomarkers to Improve Diagnosis of Renal Cell Carcinoma Subtypes
- RNA-Powered Molecular Test to Help Combat Early-Age Onset Colorectal Cancer
- Advanced Blood Test to Spot Alzheimer's Before Progression to Dementia
- Multi-Omic Noninvasive Urine-Based DNA Test to Improve Bladder Cancer Detection
- First of Its Kind NGS Assay for Precise Detection of BCR::ABL1 Fusion Gene to Enable Personalized Leukemia Treatment
- Urine Test to Revolutionize Lyme Disease Testing
- Simple Blood Test Could Enable First Quantitative Assessments for Future Cerebrovascular Disease
- New Genetic Testing Procedure Combined With Ultrasound Detects High Cardiovascular Risk
- Blood Samples Enhance B-Cell Lymphoma Diagnostics and Prognosis
- Blood Test Predicts Knee Osteoarthritis Eight Years Before Signs Appears On X-Rays
- Blood Test Accurately Predicts Lung Cancer Risk and Reduces Need for Scans
- Unique Autoantibody Signature to Help Diagnose Multiple Sclerosis Years before Symptom Onset
- Blood Test Could Detect HPV-Associated Cancers 10 Years before Clinical Diagnosis
- Low-Cost Point-Of-Care Diagnostic to Expand Access to STI Testing
- 18-Gene Urine Test for Prostate Cancer to Help Avoid Unnecessary Biopsies
- Urine-Based Test Detects Head and Neck Cancer
Channels
Clinical Chemistry
view channel
3D Printed Point-Of-Care Mass Spectrometer Outperforms State-Of-The-Art Models
Mass spectrometry is a precise technique for identifying the chemical components of a sample and has significant potential for monitoring chronic illness health states, such as measuring hormone levels... Read more.jpg)
POC Biomedical Test Spins Water Droplet Using Sound Waves for Cancer Detection
Exosomes, tiny cellular bioparticles carrying a specific set of proteins, lipids, and genetic materials, play a crucial role in cell communication and hold promise for non-invasive diagnostics.... Read more
Highly Reliable Cell-Based Assay Enables Accurate Diagnosis of Endocrine Diseases
The conventional methods for measuring free cortisol, the body's stress hormone, from blood or saliva are quite demanding and require sample processing. The most common method, therefore, involves collecting... Read moreMolecular Diagnostics
view channel
Novel Biomarkers to Improve Diagnosis of Renal Cell Carcinoma Subtypes
Renal cell carcinomas (RCCs) are notably diverse, encompassing over 20 distinct subtypes and generally categorized into clear cell and non-clear cell types; around 20% of all RCCs fall into the non-clear... Read more
RNA-Powered Molecular Test to Help Combat Early-Age Onset Colorectal Cancer
Colorectal cancer (CRC) ranks as the second most lethal cancer in the United States. Nevertheless, many Americans eligible for screening do not undergo testing due to limited access or reluctance towards... Read moreHematology
view channel
Next Generation Instrument Screens for Hemoglobin Disorders in Newborns
Hemoglobinopathies, the most widespread inherited conditions globally, affect about 7% of the population as carriers, with 2.7% of newborns being born with these conditions. The spectrum of clinical manifestations... Read more
First 4-in-1 Nucleic Acid Test for Arbovirus Screening to Reduce Risk of Transfusion-Transmitted Infections
Arboviruses represent an emerging global health threat, exacerbated by climate change and increased international travel that is facilitating their spread across new regions. Chikungunya, dengue, West... Read more
POC Finger-Prick Blood Test Determines Risk of Neutropenic Sepsis in Patients Undergoing Chemotherapy
Neutropenia, a decrease in neutrophils (a type of white blood cell crucial for fighting infections), is a frequent side effect of certain cancer treatments. This condition elevates the risk of infections,... Read more
First Affordable and Rapid Test for Beta Thalassemia Demonstrates 99% Diagnostic Accuracy
Hemoglobin disorders rank as some of the most prevalent monogenic diseases globally. Among various hemoglobin disorders, beta thalassemia, a hereditary blood disorder, affects about 1.5% of the world's... Read moreImmunology
view channel
Diagnostic Blood Test for Cellular Rejection after Organ Transplant Could Replace Surgical Biopsies
Transplanted organs constantly face the risk of being rejected by the recipient's immune system which differentiates self from non-self using T cells and B cells. T cells are commonly associated with acute... Read more
AI Tool Precisely Matches Cancer Drugs to Patients Using Information from Each Tumor Cell
Current strategies for matching cancer patients with specific treatments often depend on bulk sequencing of tumor DNA and RNA, which provides an average profile from all cells within a tumor sample.... Read more
Genetic Testing Combined With Personalized Drug Screening On Tumor Samples to Revolutionize Cancer Treatment
Cancer treatment typically adheres to a standard of care—established, statistically validated regimens that are effective for the majority of patients. However, the disease’s inherent variability means... Read morePathology
view channelHyperspectral Dark-Field Microscopy Enables Rapid and Accurate Identification of Cancerous Tissues
Breast cancer remains a major cause of cancer-related mortality among women. Breast-conserving surgery (BCS), also known as lumpectomy, is the removal of the cancerous lump and a small margin of surrounding tissue.... Read more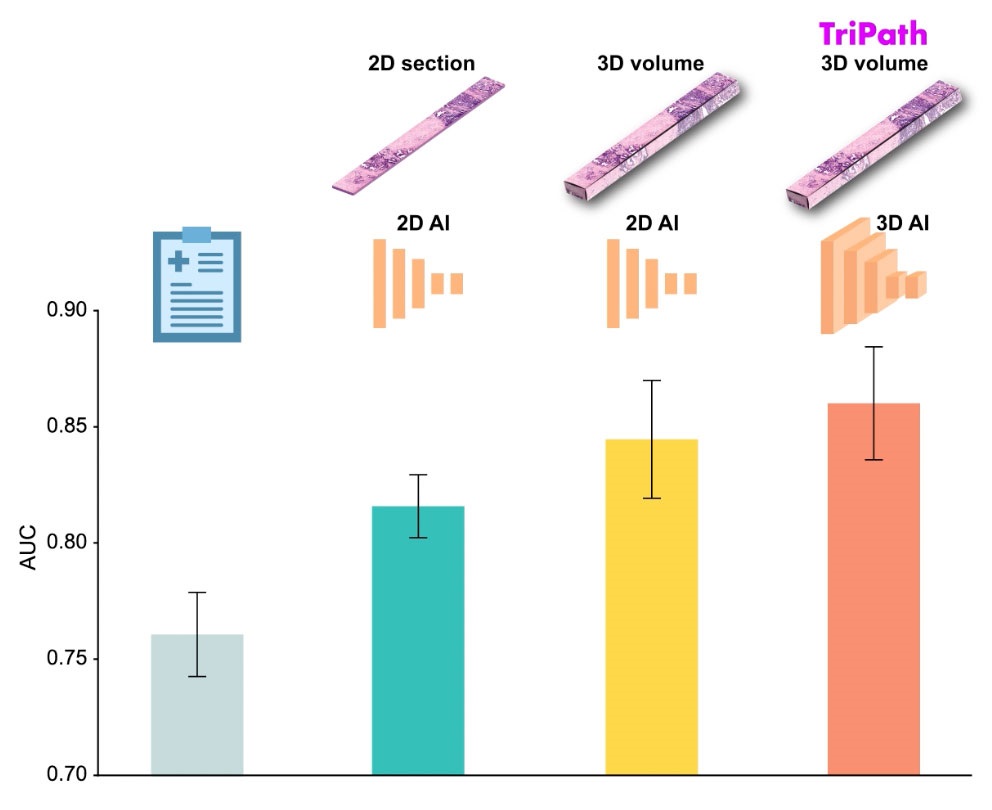
AI Advancements Enable Leap into 3D Pathology
Human tissue is complex, intricate, and naturally three-dimensional. However, the thin two-dimensional tissue slices commonly used by pathologists to diagnose diseases provide only a limited view of the... Read more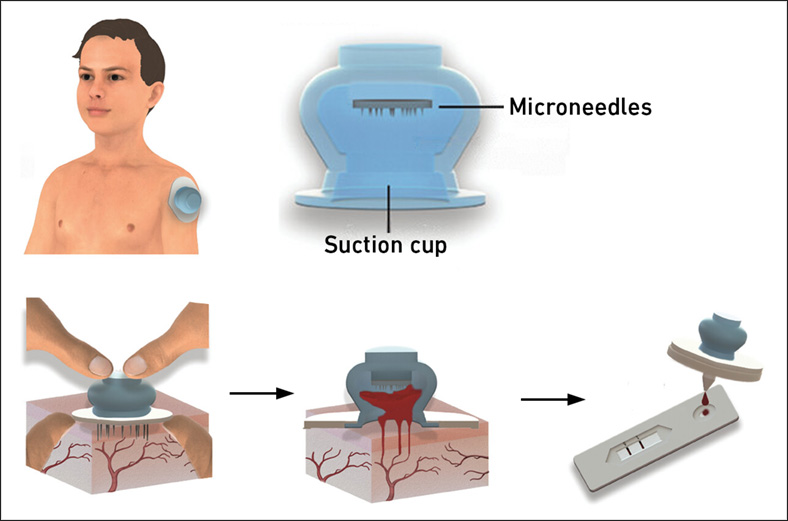
New Blood Test Device Modeled on Leeches to Help Diagnose Malaria
Many individuals have a fear of needles, making the experience of having blood drawn from their arm particularly distressing. An alternative method involves taking blood from the fingertip or earlobe,... Read more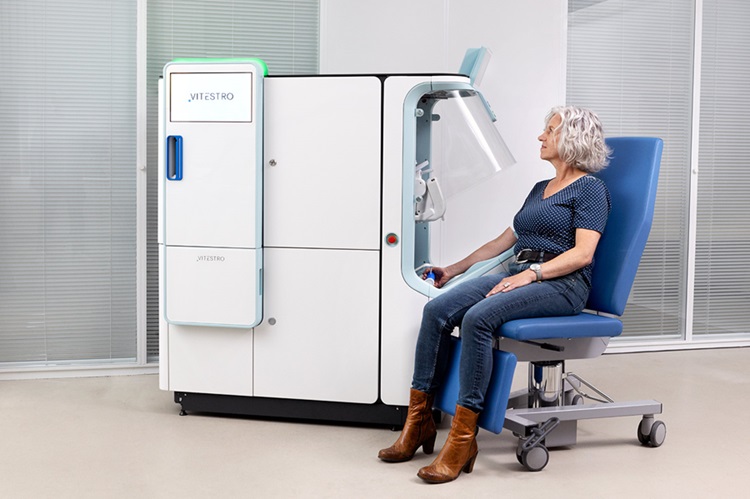
Robotic Blood Drawing Device to Revolutionize Sample Collection for Diagnostic Testing
Blood drawing is performed billions of times each year worldwide, playing a critical role in diagnostic procedures. Despite its importance, clinical laboratories are dealing with significant staff shortages,... Read moreTechnology
view channel
New Diagnostic System Achieves PCR Testing Accuracy
While PCR tests are the gold standard of accuracy for virology testing, they come with limitations such as complexity, the need for skilled lab operators, and longer result times. They also require complex... Read more
DNA Biosensor Enables Early Diagnosis of Cervical Cancer
Molybdenum disulfide (MoS2), recognized for its potential to form two-dimensional nanosheets like graphene, is a material that's increasingly catching the eye of the scientific community.... Read more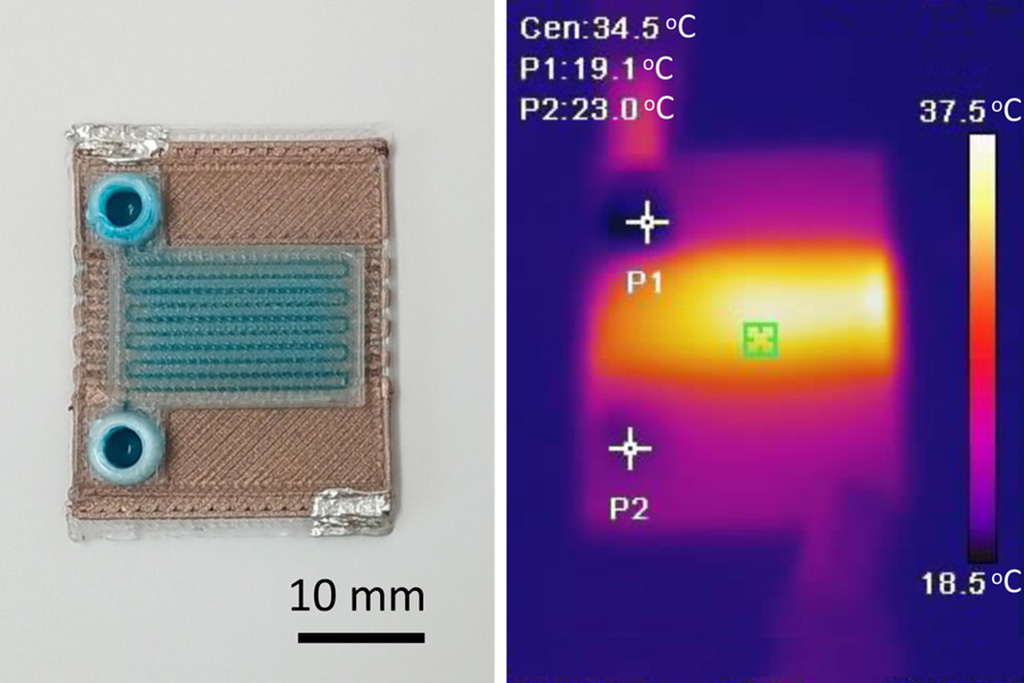
Self-Heating Microfluidic Devices Can Detect Diseases in Tiny Blood or Fluid Samples
Microfluidics, which are miniature devices that control the flow of liquids and facilitate chemical reactions, play a key role in disease detection from small samples of blood or other fluids.... Read more
Breakthrough in Diagnostic Technology Could Make On-The-Spot Testing Widely Accessible
Home testing gained significant importance during the COVID-19 pandemic, yet the availability of rapid tests is limited, and most of them can only drive one liquid across the strip, leading to continued... Read moreIndustry
view channel
Danaher and Johns Hopkins University Collaborate to Improve Neurological Diagnosis
Unlike severe traumatic brain injury (TBI), mild TBI often does not show clear correlations with abnormalities detected through head computed tomography (CT) scans. Consequently, there is a pressing need... Read more
Beckman Coulter and MeMed Expand Host Immune Response Diagnostics Partnership
Beckman Coulter Diagnostics (Brea, CA, USA) and MeMed BV (Haifa, Israel) have expanded their host immune response diagnostics partnership. Beckman Coulter is now an authorized distributor of the MeMed... Read more_1.jpg)