Urine Test Beats MRI in Identifying Prostate Cancer Upgrading During Active Surveillance
Posted on 12 May 2026
Active surveillance is common for men with low-risk prostate cancer, yet deciding when to repeat biopsy remains challenging. Prostate-specific antigen (PSA) testing and magnetic resonance imaging (MRI) may not reliably flag when indolent disease has progressed, prompting unnecessary procedures or delayed intervention. Noninvasive urine assays could help refine risk assessment and reduce biopsy burden. A new study shows that a urine test can outperform MRI and PSA for monitoring low-risk disease.
At Vanderbilt University Medical Center (Nashville, TN, USA), investigators evaluated MyProstateScore 2.0—Active Surveillance (MPS2-AS) from LynxDx, Inc. (Ann Arbor, MI, USA), a urine-based assay designed to guide repeat-biopsy decisions during active surveillance. The test was assessed in men previously diagnosed with Grade Group (GG) 1 prostate cancer. According to the study, it outperformed PSA-based testing and MRI for determining which patients need a “monitoring” biopsy and which can safely defer the procedure.
The assay uses a noninvasive urine sample to estimate the likelihood of higher-grade disease. By stratifying risk for Grade Group upgrading, it aims to maintain timely identification of cancers that merit treatment while reducing exposure to invasive sampling. The approach addresses limitations of current surveillance pathways that often default to repeat biopsies every two to three years.
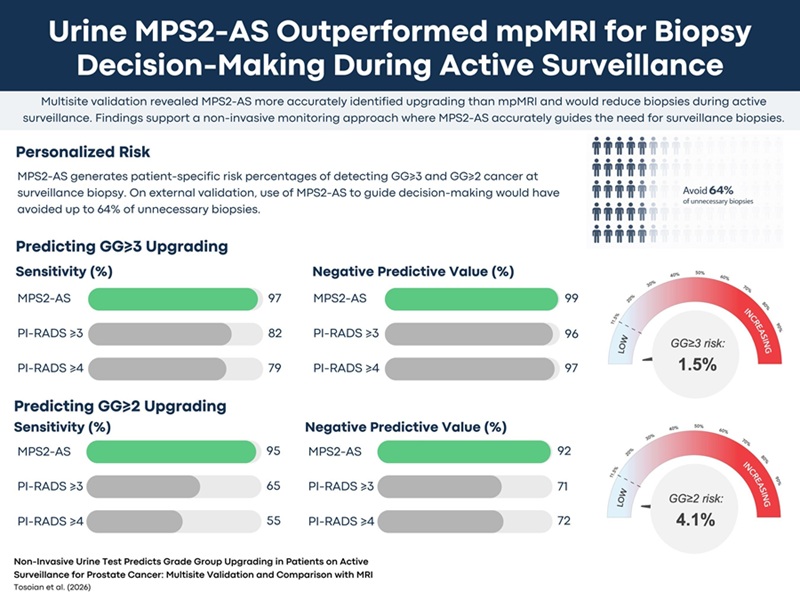
In a cohort of more than 300 patients on active surveillance for GG1 disease, MPS2-AS would have avoided up to 64% of unnecessary biopsies while preserving prompt detection of higher-grade cancers. The test correctly predicted the presence of high-grade disease (GG≥3) in 97% of cases. It achieved a 99% negative predictive value (NPV) for GG≥3 upgrading, indicating that patients with a negative result had only a 1% chance of having GG≥3 cancer found on biopsy.
Findings were published in The Journal of Urology, in a study titled “Non-Invasive Urine Test Predicts Grade Group Upgrading in Patients on Active Surveillance for Prostate Cancer: Multisite Validation and Comparison with MRI.” The research team noted that next steps include evaluating this testing approach to improve other aspects of prostate cancer care, such as detecting recurrence after treatment.
“For patients undergoing monitoring of low-grade prostate cancer, these findings suggest that use of the urine test can reduce the need for invasive biopsies without compromising prompt detection of higher-grade cancers that require treatment,” said Jeffrey Tosoian, MD, MPH, assistant professor in the Department of Urology at Vanderbilt Health.
Related Links
Vanderbilt University Medical Center
LynxDx, Inc.




.jpg)