Risk Prediction Tool Enhances Genetic Testing for Li-Fraumeni Syndrome
Posted on 28 Apr 2026
Li-Fraumeni syndrome is a rare hereditary cancer predisposition most often driven by germline mutations in the TP53 tumor suppressor gene. Determining who should receive TP53 testing remains challenging because commonly used criteria can overlook atypical or limited family histories. Many individuals referred for testing ultimately receive negative results, creating uncertainty for patients and counselors. A new study now has demonstrated that a statistical model can refine risk assessment and better align decisions with clinical judgment.
The University of Texas MD Anderson Cancer Center evaluated LFSPRO, a mathematical risk-prediction model designed to support genetic counseling for Li-Fraumeni syndrome (LFS). LFSPRO integrates complete family history to estimate the probability of carrying a TP53 mutation and to forecast risk for LFS-associated cancers. The model is intended to provide quantitative risk estimates for individuals who may benefit from testing but do not meet National Comprehensive Cancer Network (NCCN) Chompret criteria.
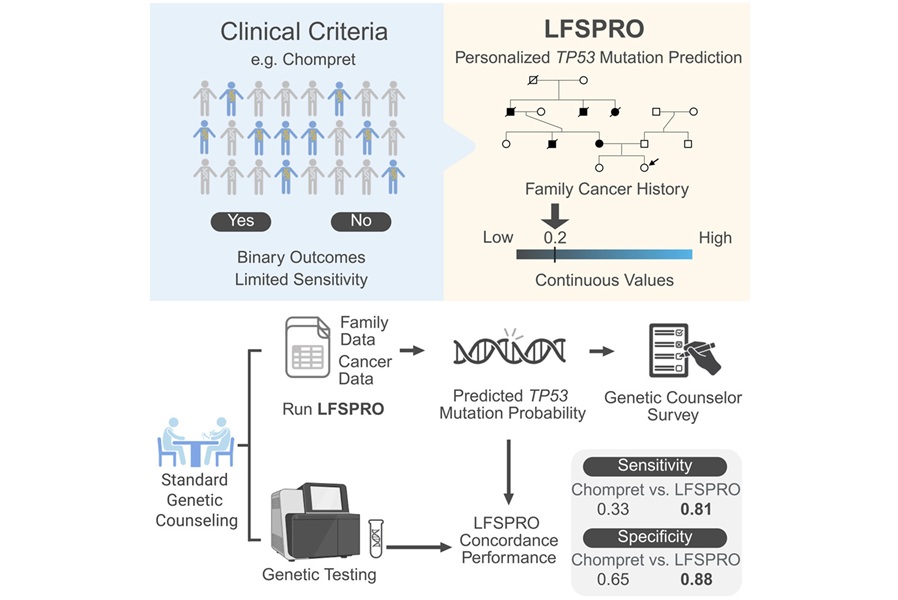
In a prospective validation study, four genetic counselors applied LFSPRO during standard sessions with 178 individuals who also underwent germline TP53 testing. The approach reflected real-world practice, with limited family history collected within 30 minutes and most counselees testing negative. The evaluation assessed how LFSPRO informed clinical decision-making and how its outputs compared with counselors’ judgments.
LFSPRO showed strong predictive performance for individual TP53 mutation probabilities and significantly outperformed the Chompret criteria across sensitivity, specificity, and positive and negative predictive values. Genetic counselors reported close concordance between LFSPRO’s estimates and their own assessments and indicated they were comfortable sharing model results with patients and families.
Findings were published in The American Journal of Human Genetics in 2026. The researchers noted that accurate identification of individuals with LFS has major implications for cancer screening and prevention, and that accessible, user-friendly tools in community settings could increase detection of at-risk individuals and enable timely surveillance strategies.
“Most risk prediction models are validated only in research-based cohorts, but our study demonstrates LFSPRO's performance in a real-world genetic counseling setting, where limited family history information is provided within 30 minutes and most counselees test negative,” said Wenyi Wang, Ph.D., professor of Bioinformatics and Computational Biology at The University of Texas MD Anderson Cancer Center. “Our model demonstrated substantially higher accuracy than current clinical guidelines and had strong concordance with genetic counselors' judgment.”
Related Links
The University of Texas MD Anderson Cancer Center