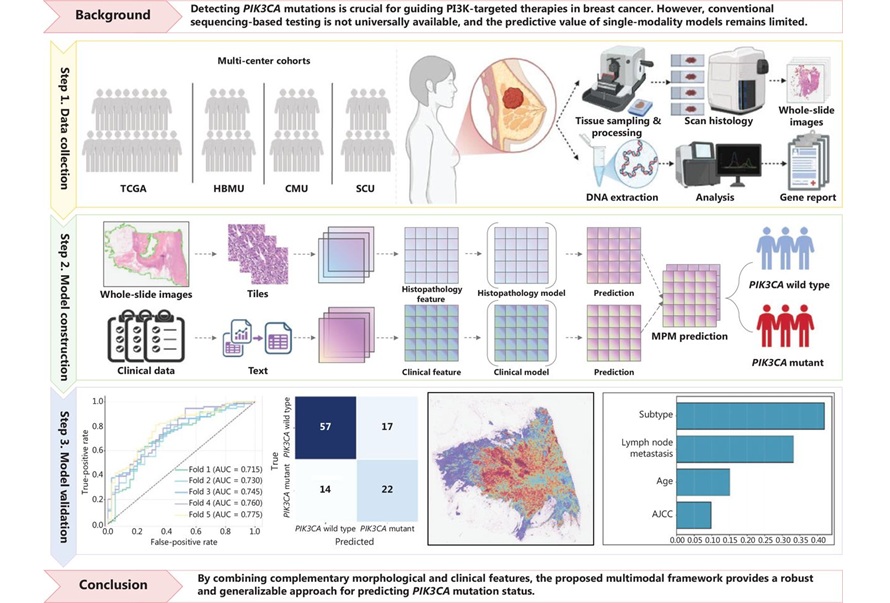
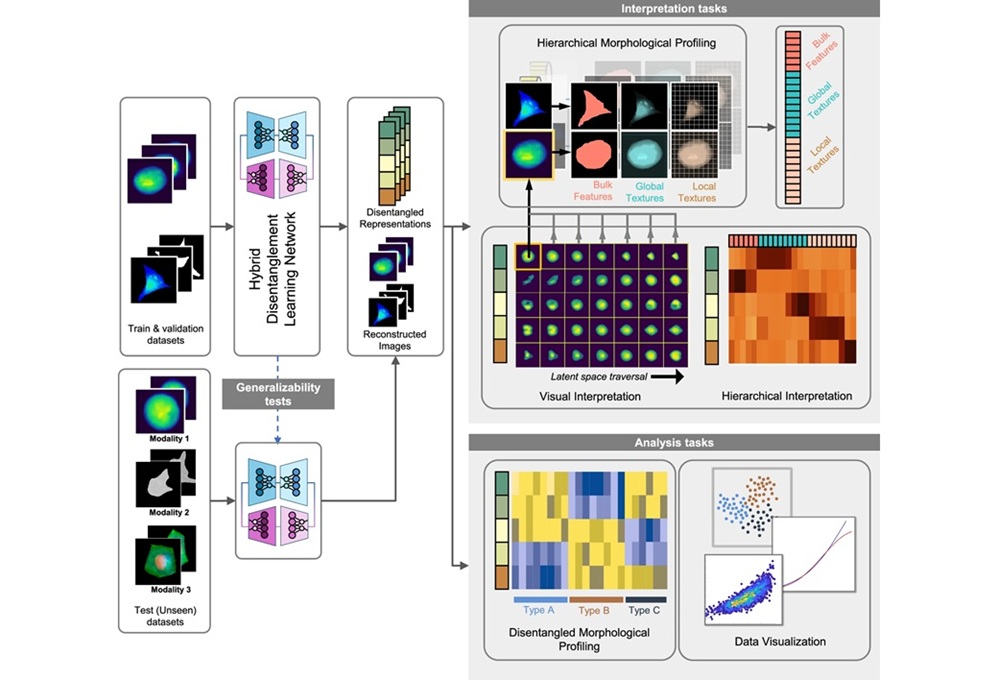
Developed Immunoassay Predicts Benefits of Anti-PD-1 Therapy in NSCLC
By LabMedica International staff writers
Posted on 12 May 2021
Lung cancer is the most common cause of cancer-related deaths worldwide. However, current targeted therapies and immunotherapies have gradually improved patient survival. Anti-programmed cell death-1 (PD-1) antibodies (Abs) are key drugs in non-small-cell lung cancer treatment; however, clinical benefits with anti-PD-1 monotherapy are limited.Posted on 12 May 2021
Notably, anti-programmed cell death-1 (PD-1) and anti-PD-ligand1 (PD-L1) antibodies (Abs) as immune-checkpoint inhibitors prolong overall survival in non-small-cell lung cancer (NSCLC), which accounts for approximately 80% of lung cancer. However, clinical benefits with anti-PD-1/PD-L1 monotherapy are limited with only 20%–30% overall response rate.

Image: The HISCL-5000 is a fully automated immunoassay system designed for fast, highly sensitive and reliable immunoassay testing (Photo courtesy of Sysmex)
Oncologists at the Kawasaki Medical School, Okayama, Japan and their colleagues obtained sera from advanced NSCLC patients, who received anti-PD-1 monotherapy as standard therapy in a first-line or later setting. Sera were collected within two months before anti-PD-1 monotherapy and were serially done after anti-PD-1 therapy, then frozen and stored at −80 °C until use. ESO-1 and XAGE1 antigen in tumor tissues were immunohistochemically stained in a previous study.
The team developed a fully automated detection system for NY-ESO-1/XAGE1 Abs using an HISCL series (Sysmex, Kobe, Japan), which performs a chemiluminescent sandwich immunoassay. Sera were diluted 400 times with appropriate solution and reacted with magnetic beads coated with recombinant NY-ESO-1 protein or synthetic XAGE1 peptide (GL Biochemistry, Shanghai, China). The investigators also determined serum Abs stability, performed analysis of interfering substances and antigen absorption tests.
The scientists reported that the HISCL system detected specific serum NY-ESO-1/XAGE1 Abs, where levels in ELISA and HISCL were highly correlated. The Ab levels in HISCL were stable at four temperatures, five freeze/thaw cycles, and long-term storage; the levels were not interfered by common blood components. The Abs levels in 15 NSCLC responders to anti-PD-1 monotherapy were significantly higher than those in non-responders and healthy donors. The AUROC was the highest (0.91) in combinatory prediction with NY-ESO-1/XAGE1 Abs.
The authors concluded that they had developed a fully automated immunoassay system, HISCL, measuring serum NY-ESO-1/XAGE1 Abs that can predict clinical benefits with anti-PD-1 monotherapy in NSCLC. Their serum biomarkers measured using HISCL are clinically meaningful, as the serum Abs obtained noninvasively were relatively stable at various conditions and could be measured easily and rapidly. The study was published on April, 15, 2021 in the journal Clinica Chimica Acta.
Related Links:
Kawasaki Medical School
Sysmex
GL Biochemistry