Ancillary Tests Developed for Salivary Gland Pathology
By LabMedica International staff writers
Posted on 03 May 2017
Salivary gland tumors represent a diverse set of tumors with a broad range of biologic behaviors, ranging from completely benign tumors to low-grade malignancies to high-grade malignancies.Posted on 03 May 2017
In addition to this wide range in clinical behavior, salivary gland tumors can show extensive morphologic overlap, so much so that, in many cases, a definitive diagnosis can be impossible without examination of the entire tumor.
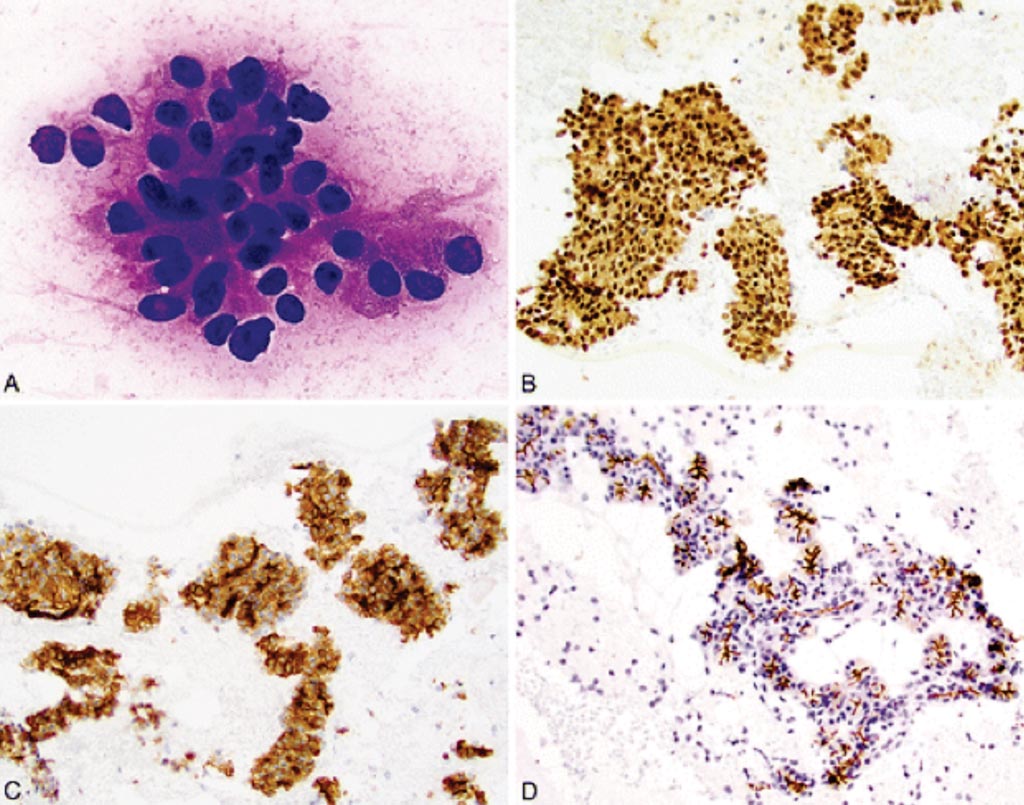
Image: An immunohistochemistry of fine needle aspirates in acinic cell carcinoma (AciCC) for SRY-related HMG-box 10 (SOX10) and 2-deoxyglucose-6-phosphate phosphatase (DOG1) (Photo courtesy of Emory University School of Medicine).
Pathologists at the Emory University School of Medicine have examined some of the new advances in salivary gland tumor pathology that have a potential for clinical impact. Ancillary testing with immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH) are improving the ability to make specific diagnoses, or at least to limit the diagnostic consideration on biopsies.
The use of IHC and/or FISH testing can be helpful in the accurate diagnosis of salivary gland tumors in both large resections and, more important, in small biopsy specimens, when combined with clinical information and morphology. These tests are even more useful when applied as part of a panel approach that incorporates tumor morphology to limit the differential diagnosis. Salivary gland tumors also occasionally show targetable molecular alterations, and combined with more-accurate diagnosis, this may allow for more-tailored treatment for the patient and the particular tumor.
Specific tumor types show different frequencies of targetable pathways; therefore, ancillary testing should be tailored to the specific diagnosis. The continuation of clinical work will probably discover more characteristic chromosomal rearrangements, useful antibodies, and targetable mutations in these rare tumors. The study was published in the March 2017 issue of the journal Archives of Pathology & Laboratory Medicine.














