Reliability Reevaluated for Bladder Cancer Test Prognosis
By LabMedica International staff writers
Posted on 08 Nov 2016
A blood test that had shown promise in predicting how cancer will progress and what treatments will be most effective for a given patient may not be reliable for either, as results from previous studies are threatened by methodological limitations.Posted on 08 Nov 2016
Preoperative risk stratification is a major challenge in bladder cancer (BC) and a robust biomarker is needed. One emerging candidate is the neutrophil-to-lymphocyte ratio (NLR). NLR is easily calculated from a complete blood count (CBC) and is felt to reflect the systemic inflammatory state.
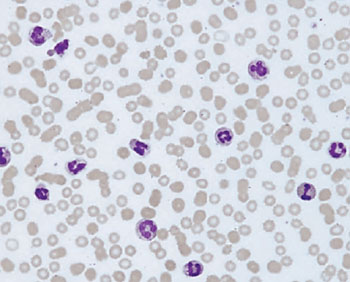
Image: A blood film from a patient with bladder cancer showing increased number of neutrophils and band cells (Photo courtesy of MPH).
Medical scientists at the University of Pennsylvania (Philadelphia, PA, USA) and their colleagues analyzed data that was collected in real-time during a prospective clinical trial, making it the first study of NLR in bladder cancer not to rely on observational data. The study analyzed SWOG 8710, which was a randomized Phase III trial of 317 patients with muscle-invasive bladder cancer. All patients were treated with radical cystectomies. Half had pre-surgery chemotherapy, while the other half did not.
Of the 317 total patients, the team identified 230 for a prognostic analysis to see if NLR could serve as a predictor of how long patients would live after curative treatment. They identified 263 others for a predictive analysis to see if NLR could tell which patients would respond to chemotherapy. There was a median follow-up of 18.6 years. For the prognostic analysis, NLR was not a significant factor in overall survival. The important factors were age and whether the patient received pre-surgery chemotherapy. For the predictive analysis, NLR did not predict which patients benefitted from chemotherapy.
Eric Ojerholm, MD, the lead author of the study, said, “The trial we used has a few big advantages to study NLR. First, baseline blood samples were collected as part of the trial protocol. Second, the study’s long-term follow-up gave us adequate ‘statistical power,’ meaning that if NLR really was a biomarker, then we should be able to detect it. Third, the trial randomly assigned some patients to receive pre-surgery chemotherapy. This allowed us to test NLR both as a prognostic and predictive biomarker.” The study was published on October 27, 2016, in the journal Cancer.
Related Links:
University of Pennsylvania














