Commercial Assay Aids Physicians in Tamoxifen Drug Monitoring
By LabMedica International staff writers
Posted on 30 Dec 2015
A laboratory developed test could help assess levels of several tamoxifen drug metabolites in a blood specimen which is important as five years of therapy with tamoxifen has been the standard of care for women with early stage, estrogen-receptor-positive (ER+) breast cancer following "curative" surgery. Posted on 30 Dec 2015
Tamoxifen, taken for five years, reduces the risk of developing invasive breast cancer by about 50% in postmenopausal women who are at increased risk of getting the disease. However, compliance is an issue and patients could be further helped from precise prescribing as side effects from Tamoxifen are known to include mood swings, depression, and loss of libido, cataracts, blood clots, strokes, bone loss and the increase risk of endometrial and uterine cancer.
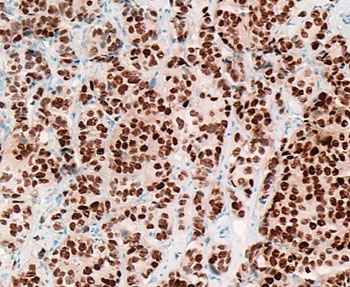
Image: Immunohistochemistry of an estrogen receptor-positive breast cancer surgical pathology specimen; most of the cancer cell nuclei stain strongly positive (dark brown) for estrogen receptor (ER) (Photo courtesy of Dr. Ronald S. Weinstein, MD).
Medical experts from Quest Diagnostics (Madison, NJ, USA) show how the laboratory developed test could help assess levels of several tamoxifen drug metabolites in a blood specimen. The scientists evaluated the blood serum from 155 women who were on 20 mg/day tamoxifen for more than six months. The patient population exhibited a wide range of concentrations of tamoxifen and its metabolites during therapy, reflecting individual differences in the ability to metabolize the drug.
The test is based on a tandem mass spectrometry, (LC-MS/MS) a technique used by world-class laboratories to measure hormones and vitamin D, the test allows physicians, for the first time, to employ a commercially available therapeutic drug monitoring approach for their tamoxifen-treated patients. The test may also help physicians assess noncompliance with tamoxifen therapy, a common problem given side effects such as weight gain or loss.
Nigel Clarke, PhD, coauthor and a senior scientific expert responsible for technology innovation for Quest Diagnostics, said, “When people think of personalized medicine or precision medicine they often think of genetic testing to assess a drug against a gene variant. But a growing area in personalized medicine is the use of qualitative enzyme or drug metabolite testing to assess the biological impact of a drug rather than the indirect influence of genes.” The study was presented on December 11, 2015, at the San Antonio Breast Cancer Symposium held in San Antonio (TX, USA).
Related Links:
Quest Diagnostics














