Noninvasive Urine Test Predicts Recurrence After BCG in Bladder Cancer
Posted on 09 Apr 2026
Bladder cancer is among the most common malignancies in the United States and frequently recurs even when diagnosed at the non‑muscle invasive stage (NMIBC). After transurethral resection, many patients receive Bacillus Calmette‑Guérin (BCG), an intravesical immunotherapy, yet clinicians lack precise tools to determine who truly harbors residual disease. Shortages and side effects of BCG intensify the need for accurate, noninvasive surveillance beyond cystoscopy. A new study shows that analyzing tumor DNA in urine can clarify treatment benefit and stratify recurrence risk after surgery and immunotherapy.
Stanford University investigators developed a field‑effect‑informed urine liquid biopsy that quantifies tumor DNA while correcting for age‑related background mutations in normal urothelium. The team observed that even healthy bladders can carry cancer‑related mutations, a phenomenon they termed clonal cystopoiesis. To address this confounder, they built a statistical method that filters these signals so the assay more accurately detects minimal residual disease.
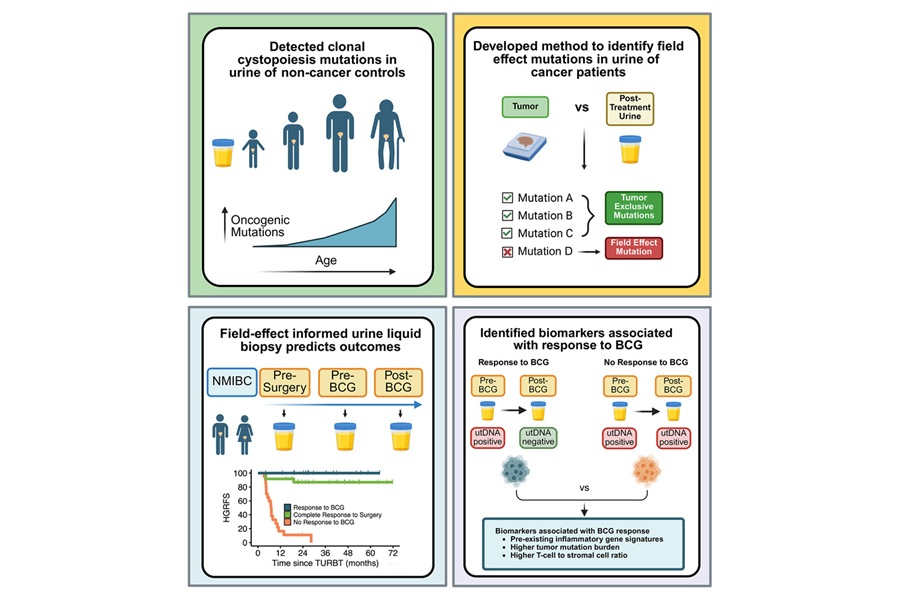
Applied prospectively to patients undergoing tumor resection followed by BCG, the refined assay was strongly predictive of outcome. Detectable tumor DNA after completing BCG was associated with an almost certain risk of recurrence, whereas clearance of tumor DNA correlated with excellent outcomes. In many cases the assay indicated elevated risk even when cystoscopy appeared normal, suggesting earlier detection than routine surveillance.
Serial urine testing before surgery, after surgery, and after immunotherapy revealed three molecular response patterns: surgery responders with disappearance of tumor DNA after resection; BCG responders with decreases after intravesical therapy; and non‑responders with persistence or increases despite BCG. Correcting for the field effect was essential to separate these categories and improved the specificity of urine tumor DNA detection. Analyses further suggested that tumors responding to BCG tended to have higher mutation burdens and pre‑existing immune activity, distinct from biology linked to surgical response.
The study, led by the Stanford Departments of Urology and Radiation Oncology in collaboration with the Stanford Cancer Institute, was published in Cell on February 19, 2026. According to the authors, a field‑effect‑informed urine test could help spare patients who are molecularly cured after surgery, prioritize BCG for those most likely to benefit, escalate treatment earlier for those at highest risk, and reduce unnecessary procedures caused by false positives. The team also noted that similar field effects are seen in other epithelia, indicating that adjusting for age‑related background mutations may be important as liquid biopsies expand across cancer types and biofluids.
"These kinds of predictive biomarkers are critical. We have new treatments that are costly and carry risk of side effects. We would love to personalize therapy to ensure each patient receives the best treatment for their individual cancer," said Eila Skinner, MD, the Thomas A. Stamey Research Professor of Urology and Chair of Stanford's Department of Urology.
Related Links
Stanford Medicine