New Imaging Approach Could Help Predict Dangerous Gut Infection
Posted on 10 Mar 2026
Clostridioides difficile infections affect roughly half a million people in the United States each year and are a leading cause of infectious diarrhea in healthcare settings. The bacterium can trigger severe intestinal inflammation, recurring illness, and life-threatening complications, particularly in older adults. Despite available treatments, controlling C. diff infections remains difficult because the bacterium can survive harsh conditions and frequently reappears after treatment. Researchers are now investigating the bacterium at multiple biological levels to uncover vulnerabilities that could lead to improved prevention and treatment strategies.
Scientists at Tufts University School of Medicine (Boston, MA, USA) are studying how C. diff behaves inside the body and how its spores reactivate to cause infection. C. diff infections typically occur after antibiotics disrupt the natural gut microbiome, allowing the bacterium to colonize the colon and release toxins that damage intestinal tissues. To better understand how the infection progresses, researchers developed an imaging technique that tracks individual bacterial cells inside infected tissue. Using fluorescent markers that highlight gene activity, the team observed where C. diff cells were located and which cells activated toxin production during infection.
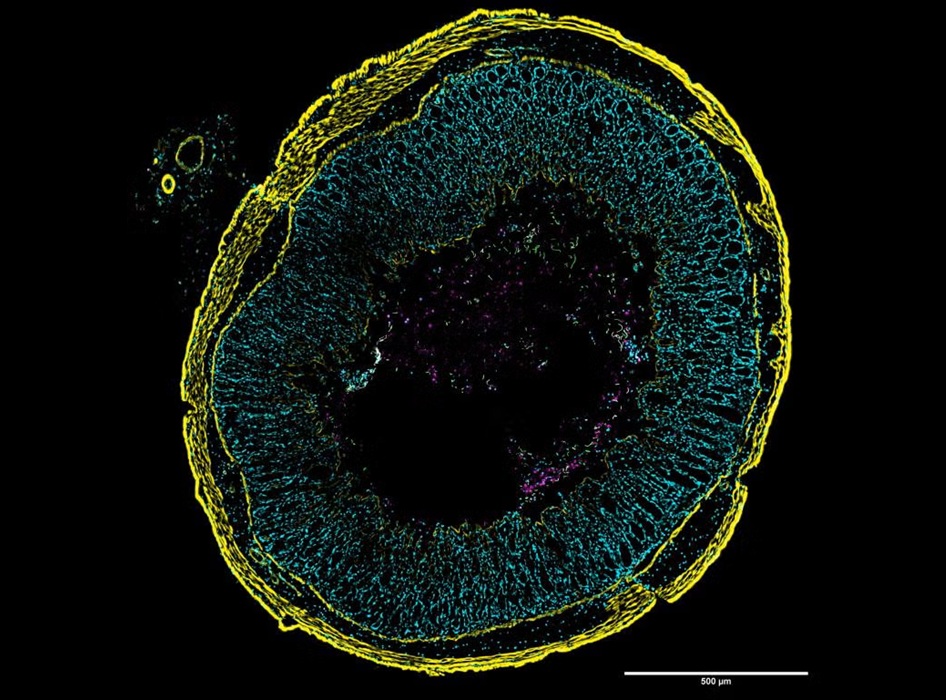
The analysis revealed that C. diff bacteria can spread throughout the gut and reside closer to the intestinal lining than previously thought. Surprisingly, only a small subset of bacterial cells produced toxins at any given time, suggesting that disease severity may be driven by specific subpopulations rather than overall bacterial numbers. The study, published in Nature Communications and PLOS Biology, also identified unusual filament-like shapes formed by highly toxin-producing strains during early infection stages, indicating that these cells may respond differently to environmental stress.
Researchers also examined how C. diff spores transition from a dormant state into active bacteria. Unlike most spore-forming bacteria, C. diff responds to bile acids and other digestive signals to trigger germination. The team identified two proteins—CspC and CspA—that work together as a signaling hub controlling this activation process. Structural analysis showed that this protein complex regulates how sensitive spores are to environmental signals that trigger growth.
These findings provide a clearer understanding of how C. diff infections develop, spread, and reactivate. By identifying the molecular mechanisms that control toxin production and spore germination, researchers hope to develop targeted treatments that block infection without disrupting beneficial gut bacteria. Future research will continue exploring unique biological processes in C. diff that could reveal new drug targets and help clinicians better predict which patients are at risk of severe or recurrent infection.
“The hope is the aspects that make C. diff unique—how it spreads, reproduces, and damages tissue—will allow researchers to design ways to target it much more specifically, while keeping the rest of the gut microbiome healthy and intact,” said associate professor Aimee Shen.
Related Links:
Tufts University