Low-Cost Paper-Based Assay Enables POC Testing for Cervical Precancer
Posted on 29 Jan 2025
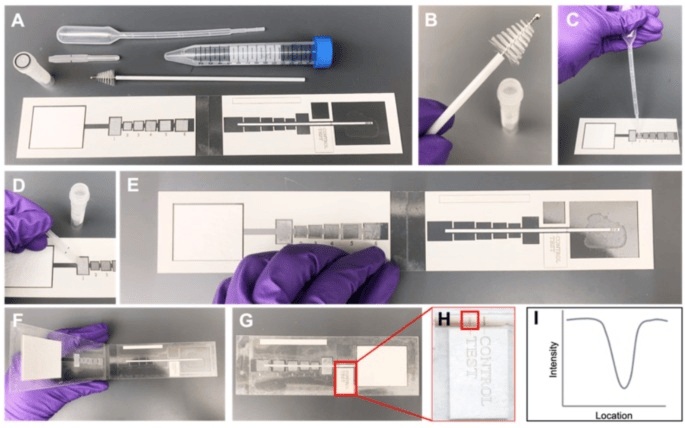
Cervical cancer is preventable through screening and treatment of cervical precancer, yet it remains a significant global challenge, particularly in low- and middle-income countries (LMICs), where the disease burden is disproportionately high. The lack of affordable, user-friendly screening and diagnostic tests exacerbates this issue. Many commercially available tests are unsuitable for LMICs due to resource limitations. For example, HPV mRNA and oncoprotein tests, which have high specificity for cervical precancer and cancer, require complex sample preparation and costly equipment. To address these challenges, researchers have developed a paper-based HPV E7 oncoprotein assay that is suitable for point-of-care use, involves only five simple steps, and does not require any instrumentation.
The HPV E7 oncoprotein assay, developed by researchers at Rice University (Houston, TX, USA), is a low-cost, sample-to-answer test that builds on their previous work. This paper-based enzyme-linked immunoassay (ELISA) is highly sensitive due to signal amplification and includes just five simple steps, including sample preparation and lysis. The point-of-care assay detects cervical precancerous lesions with minimal user input, instrumentation, or infrastructure, making it ideal for use in resource-limited settings. Additionally, the successful lyophilization of reagent pads ensures a streamlined 15-minute workflow, with results available in one hour. The test costs less than USD 1 per test with small-scale manufacturing, and USD 1.47 when including the cervical collection brush, lysis tube, and disposable pipettes.
The assay has been validated using HPV16, 18, and 45 cellular samples and in a pilot clinical study, demonstrating a sensitivity of 100% and specificity of 90%. While larger-scale validation is still needed, the study, published in Scientific Reports, confirms that the HPV E7 oncoprotein test performs well with clinical samples, detecting CIN 2 + pathology with high sensitivity and specificity. This assay could serve as a follow-up test for women who test positive for high-risk HPV DNA, helping to identify those at higher risk of preinvasive disease. It could also potentially be used as a standalone test in same-day screen-and-treat programs after further clinical validation. The paper-based, low-cost test offers the potential to screen, diagnose, and treat women for CIN 2 + lesions in one visit, reducing loss to follow-up and preventing overtreatment in already resource-limited settings.














