New POC Malaria Test 12 Times Faster Than Currently Available Laboratory-Based Tests
Posted on 01 Mar 2024
Malaria continues to pose a major global health threat, with approximately 247 million cases and over 600,000 deaths annually, predominantly in sub-Saharan Africa. Particularly alarming is cerebral malaria, a severe form of the disease, which has a high mortality rate, especially among children under five. The existing rapid diagnostic tests (RDTs) for malaria offer a basic positive or negative result, but often fail to detect asymptomatic infections and lack the sensitivity required for early detection of severe cases. More sensitive molecular assays that exist are costly, time-consuming, and need specialized skills and equipment, rendering them unsuitable for widespread application in areas with limited resources. In light of these challenges, researchers have now developed a new test for diagnosing malaria that is both rapid and accurate. This POC rapid malaria test offers a significant improvement over traditional tests and will be especially beneficial for rural areas with limited healthcare facilities.
Researchers at Rice University (Houston, TX, USA) developed a microfluidic point-of-care (mPOC) immunoassay for quantifying a malaria parasite biomarker, Plasmodium falciparum histidine-rich protein 2 (PfHRP2), in whole blood. This device provides dual diagnostic modes to detect PfHRP2 at low and high concentrations, making it versatile for various diagnostic needs, such as identifying asymptomatic infections and predicting disease progression. The test produces results in just 15 minutes, which can be easily accessed via a smartphone app developed by the researchers. Field tests in Malawi revealed that the mPOC immunoassay matches the accuracy of the standard PfHRP2 enzyme-linked immunosorbent assay (ELISA), but is 12 times quicker and simpler to operate. This advancement in malaria diagnostics, especially for cerebral malaria, holds the promise of early detection and prompt treatment of severe cases, potentially saving numerous lives.
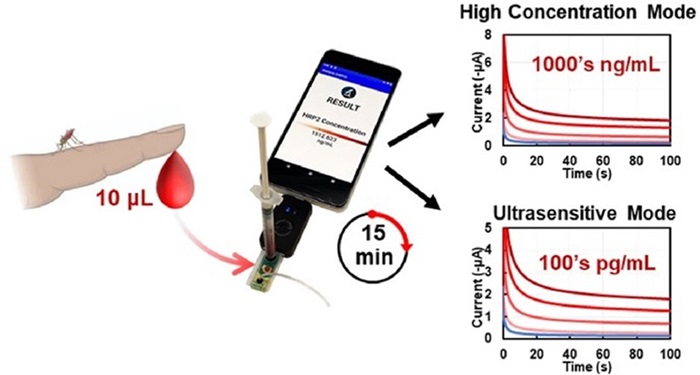
“The mPOC immunoassay was designed to be simple, accurate and field-deployable, making it suitable for use in rural and remote health centers in sub-Saharan Africa,” said mechanical engineer Peter Lillehoj of Rice’s Brown School of Engineering who led the research team. “Unlike traditional tests, this device does not require plasma separation, pipetting, complicated sample processing or long incubations, making it easy to use even by minimally trained health care providers.”
“In areas with limited access to health care facilities, our test could be a game-changer,” added Lillehoj. “It can help health care providers quickly identify and treat severe cases, potentially saving lives. By enabling early detection and appropriate management of malaria cases, we can reduce the burden of the disease and improve patient outcomes in Africa and beyond.”
Related Links:
Rice University














