Noninvasive Biomarker Tests Could Eliminate Painful Liver Biopsies
Posted on 08 Sep 2023
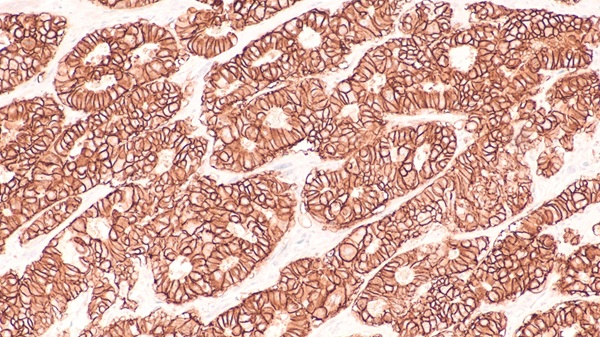
Metabolic dysfunction-associated steatohepatitis (MASH), previously known as NASH, is a severe illness. Once the liver begins to scar, the risk of cirrhosis, liver cancer, and death significantly increases. Unfortunately, symptoms of MASH often do not appear until the disease reaches its advanced stages, typically when cirrhosis has developed. Currently, the sole method to diagnose MASH in patients is through liver biopsies. However, this approach offers insights from only a small portion of the liver and fails to provide an accurate overall assessment of the patient's condition. Liver biopsies are also painful, invasive, and expensive for patients. Despite some companies developing blood and imaging biomarker tests, none have met the stringent requirements for FDA regulatory approval. Now, a consortium dedicated to investigating noninvasive tests for liver disease has achieved a significant milestone by demonstrating the effectiveness of five such tests, moving closer to regulatory approval.
The research to evaluate blood and imaging biomarker tests for liver disease is part of NIMBLE – the Noninvasive Biomarkers of Metabolic Liver Disease project – was led by the Virginia Commonwealth University (VCU, Richmond, VA, USA). The NIMBLE biomarker tests were rigorously compared to established measures, including the FIB-4 index used to assess liver health. Any biomarker test that matched or exceeded the performance of existing lab tests was assessed for its potential use in diagnosing MASH and related conditions. Over 1,000 patients participated in this research. These five biomarker tests hold promise for diagnosing MASH and related conditions, providing a noninvasive alternative to liver biopsies. This development is expected to encourage patient participation in future clinical trials, which would otherwise necessitate invasive liver biopsies. Additionally, the use of noninvasive blood tests could significantly reduce the cost of conducting such studies, thus fostering greater interest in drug development. Equally important, these tests could be easily administered in a doctor's office.
“This brings us a step closer to having simple blood-based tests that can be ordered virtually in any clinical setting and will provide access to care to patients,” said Arun Sanyal, M.D., a professor at the VCU School of Medicine. “Such a step will facilitate our ability to identify those most at risk of outcomes and target them for therapy. It will also serve as a foundation for prognostic, disease monitoring and treatment-response biomarker development.”
Related Links:
VCU