Tumor Biomarker Panel Improves Identification of Candidates for Lung Cancer Screening
By LabMedica International staff writers
Posted on 17 Jan 2022
Use of a blood-based tumor biomarker panel in combination with modeling according to results of PLCOm2012 (Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial) significantly improved risk assessment for lung cancer screening.Posted on 17 Jan 2022
Investigators at the University of Texas M. D. Anderson Cancer Center (Houston, USA) hypothesized that a combination of a blood-based four-marker protein panel combined with the (PLCO)m2012 lung cancer prediction model would better identify individuals for lung cancer screening compared with current U.S. Preventive Services Task Force (USPSTF) criteria.
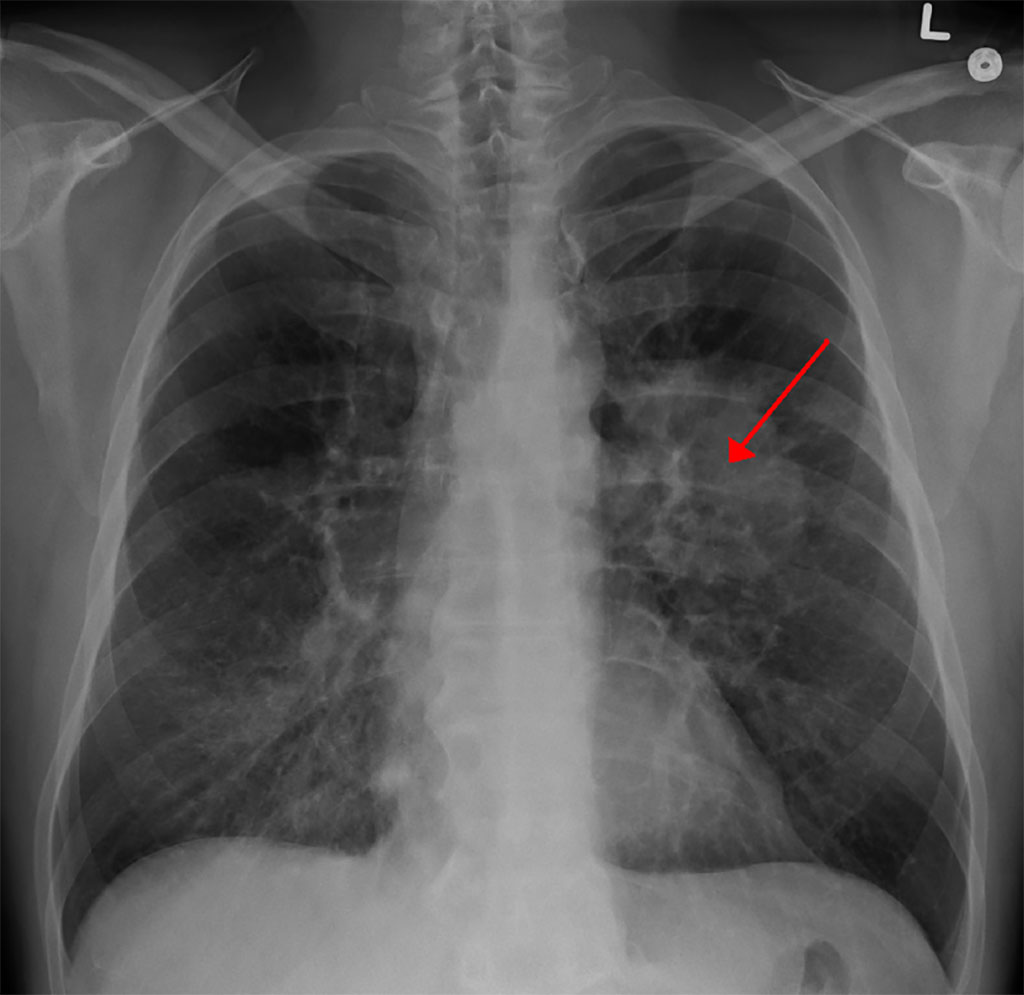
Image: A chest X-ray showing a tumor in the lung (marked by arrow) (Photo courtesy of Wikimedia Commons)
To test the hypothesis, the investigators carried out a validation study using PLCO Cancer Screening Trial data and biospecimens to evaluate the performance of a four-marker protein panel (4MP) consisting of (1) the precursor form of surfactant protein B, (2) cancer antigen 125, (3) carcinoembryonic antigen, and (4) cytokeratin-19 fragment and compared results to current USPSTF screening criteria.
The validation study included analysis of 1,299 blood samples collected from 552 individuals who developed lung cancer and 8,709 samples collected from 2,193 people who did not develop the disease. Results revealed that the 4MP alone yielded an area under the receiver operating characteristic curve of 0.79 for case sera collected within one year preceding diagnosis and 0.74 among the entire specimen set. The combined 4MP + PLCOm2012 model yielded an area under the receiver operating characteristic curve of 0.85 for case sera collected within one year preceding diagnosis. The benefit of the 4MP in the combined model resulted from improvement in sensitivity at high specificity. Compared with the USPSTF2021 criteria, the combined 4MP + PLCOm2012 model exhibited statistically significant improvements in sensitivity and specificity.
Among PLCO participants with a history of more than 10 smoking pack-years, the 4MP + PLCOm2012 model would have identified for annual screening 9.2% more lung cancer cases and would have reduced referral by 13.7% among non-cases compared with USPSTF2021 criteria.
“We recognize that a small percentage of people who are eligible for lung cancer screening through an annual low-dose CT scan are actually getting screening. Moreover, CT screening is not readily available in most countries. So, our goal, for many years, has been to develop a simple blood test that can be used first to determine need for screening and make screening for lung cancer that much more effective,” said senior author Dr. Samir M. Hanash, professor of clinical cancer prevention at the University of Texas M. D. Anderson Cancer Center. “Our study shows for the first time that a blood test could be useful to determine who may benefit from lung cancer screening. A blood test would identify people who could benefit from lung cancer screening but are not eligible today. Tens of millions of people worldwide could benefit from lung cancer screening. If you can improve screening eligibility by even 5%, that is incredibly impactful.”
The study was published in the January 7, 2022, online edition of the Journal of Clinical Oncology.
Related Links:
University of Texas M. D. Anderson Cancer Center