Saliva-Based Assay Developed for HPV-Driven Oropharyngeal Cancer
By LabMedica International staff writers
Posted on 04 Jun 2020
The incidence of high-risk human papillomavirus (HR-HPV−16, −18, −33) driven oropharyngeal cancer (OPC) is rapidly increasing in developed countries. HPV-driven OPCs have surpassed cervical cancer as the most common HPV-driven cancer in the USA.Posted on 04 Jun 2020
Oropharyngeal cancer caused by human papillomavirus (HPV) is a rising global concern. Early lesions are small and are often located in difficult to access areas (such as the crypts of the tonsils or base of tongue). Unlike cervical cancer, there is no standard or routine screening program for HPV-driven OPC.
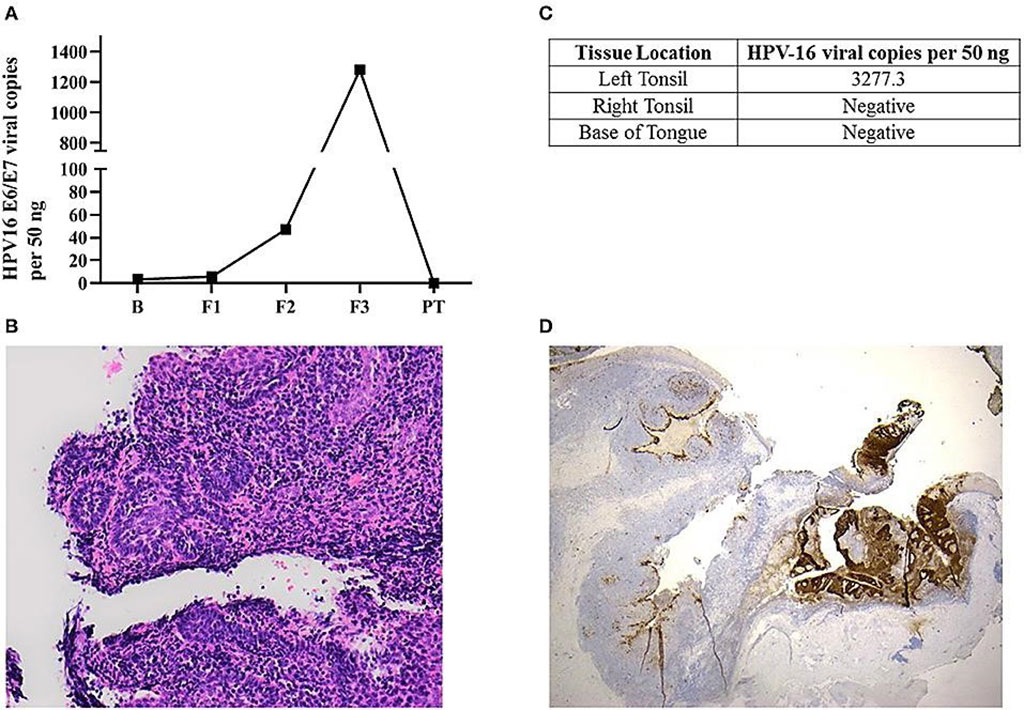
Image: Left panel: Histological sections of the left tonsillar tissue found a 2 mm non-keratinizing squamous cell carcinoma, with focal stromal invasion comparing non-affected area in the left tonsil (Photo courtesy of Queensland University of Technology).
Biomedical Scientists at the Queensland University of Technology (Brisbane, Australia) and their colleagues recruited 650 cancer-free healthy individuals with sufficient amount of DNA were tested for oral HPV-16 DNA. All were from the Queensland Region, Australia and studied between May 2016 and October 2017. Of these three have been identified to have persistent oral HPV-16 DNA infection.
From a single individual salivary oral rinse samples were collected at baseline, 6, 12, 36 month, and 2 weeks after his bilateral tonsillectomy. Total DNA was extracted from salivary oral rinse and tonsillar tissue samples using the QIAmp DNA Mini Kit (Qiagen, Germantown, MD, USA). For detection of HPV-16 genotyping, the qPCR assay targeting the opening reading frame (ORF) region of HPV16 E6/7 was carried out with the QuantStudio 7 Flex Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). The team also performed immunochemistry, DNA genotyping and viral load studies and histology.
The team observed that the HPV-16 biomarker fell to undetectable levels in saliva after surgeons removed cancerous tumors from a high-risk study participant that they had been tracking for three years. Although the report does not provide enough evidence that the test can be used as a screening tool, it demonstrates that a screened individual can receive significantly less morbid treatment than would be required for the standard presentation at a more advanced stage. The pattern of salivary HPV-DNA detection in this case demonstrated an exponential upward trend with the titer at first sample being 3.43 copies per 50 ng and the final titer before surgery of 1281.7 viral copies per 50 ng.
The authors concluded that their report support the value of a salivary oral rinse test as a potential screening tool. Unlike previously published work, the study is the first to demonstrate that continuous monitoring of HPV-16 DNA in salivary oral rinse samples can detect occult OPC. The study was published originally March 31, 2020 in the journal Frontiers in Oncology.














