Liquid Biopsy Lung Cancer Screening Method Developed
By LabMedica International staff writers
Posted on 09 Apr 2020
Genomic blood tests for cancer screening and early detection have become the focus of attention in the molecular diagnostic space, though most activity so far has been either toward pan-cancer screening tools, or in a few other specific tumor types like colorectal cancer, where tests hope to vie against colonoscopy and existing stool-based methods.Posted on 09 Apr 2020
Radiologic screening of high-risk adults reduces lung-cancer-related mortality; however, a small minority of eligible individuals undergo such screening in the USA. The availability of blood-based tests could increase screening uptake. A novel circulating tumor DNA (ctDNA) detection assay has been developed that could help physicians screen individuals at risk for lung cancer.
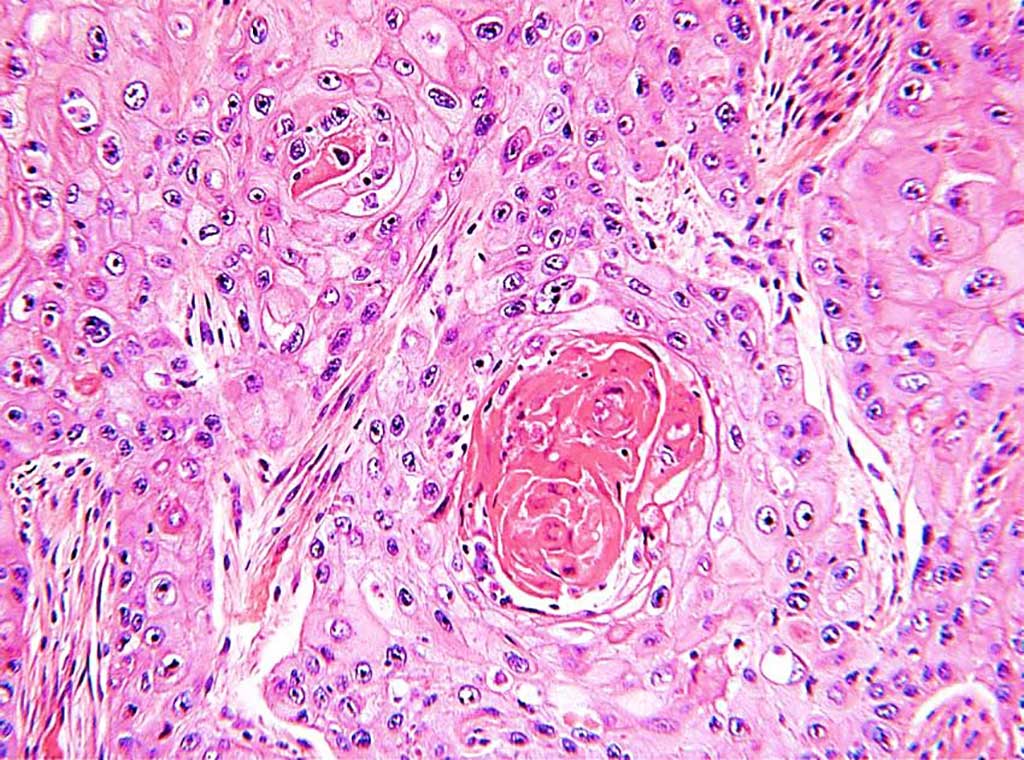
Image: Histological sections of a moderately well differentiated squamous cell carcinoma of the lung showing infiltrating sheets and tongues of malignant squamous cells with whorls of keratin (Photo courtesy of Department of Health Western Australia).
A large team of scientists at Stanford University (Stanford, CA, USA) and their colleagues have described a method, called Lung-CLiP (lung cancer likelihood in plasma), involves targeted sequencing of cell-free DNA (cfDNA) from plasma and matched white blood cell DNA to assess copy number and single nucleotide variants, coupled with a machine learning model that estimates the probability that a cfDNA mutation is tumor-derived. The estimate is based on biological and technical features specific to each variant, such as background frequency, cfDNA fragment size, the gene affected, and the likelihood of clonal hematopoiesis.
The team first trained and optimized Lung-CLiP in an initial sample of 104 patients with early stage non-small cell lung cancer and 56 matched controls. When they then applied it to an independent set of validation samples (46 cases and 48 risk-matched controls), the test was able to discriminate early-stage lung cancer patients with sensitivity and specificity levels that the authors believe suggest a significant benefit to the clinic: depending on where they set their specificity threshold, the method could achieve 63% sensitivity for stage I tumors and up to 75% sensitivity in detecting patients with stage III disease. Setting their cutoff at 98% specificity, the investigators found that Lung-CLiP detected 41% of patients with stage I disease, 54% of patients with stage II disease and 67% of those with stage III disease.
Ash Alizadeh, MD, PhD, an associate professor of Oncology and a senior author of the study, said, “Lung-CLiP could help increase the rate of early detection. This would be analogous to how stool-based testing proposes to improve screening for colorectal cancers, especially in populations where adoption of colonoscopy is lower than currently recommended.” The study was published on March 25, 2020 in the journal Nature.
Related Links:
Stanford University














