Liquid Biopsy Test Launched for NRAS Mutations
By LabMedica International staff writers
Posted on 20 Sep 2017
The NRAS gene encodes for the N-Ras protein, which is involved in communicating signals from the exterior of the cell to the cell's interior (called signal transduction). These signals provide instructions for a cell to proliferate or differentiate.Posted on 20 Sep 2017
Mutations in the NRAS gene lead to dysregulated N-Ras protein function, resulting in uncontrolled cell growth and division. NRAS mutations are associated with the development of several types of cancer, such as melanoma (15%-20% of cases), thyroid carcinoma (6%), colorectal cancer (1%-6%), and lung cancer (1%).
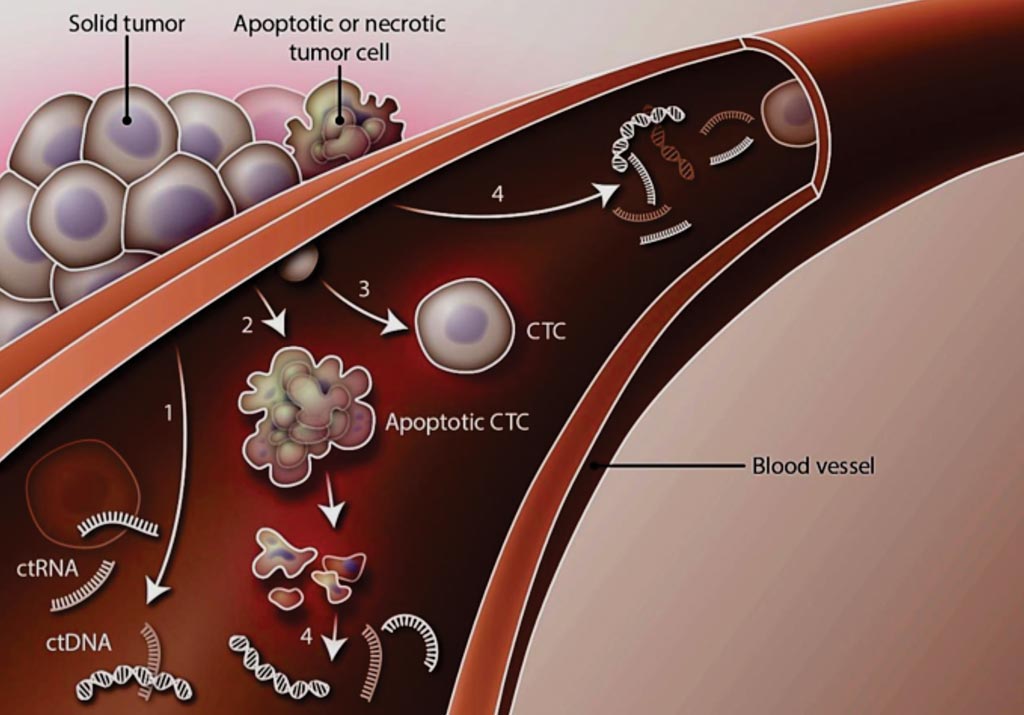
Image: Liquid Biopsy: Solid tumor masses may shed (1) circulating tumor DNA (ctDNA) and circulating tumor RNA (ctRNA), (2) apoptotic circulating tumor cells (CTCs), or (3) intact CTCs into the circulation. In addition, ctDNA and ctRNA may enter the bloodstream after apoptosis of circulating and non-circulating tumor cells (4) (Photo courtesy of H. McDonald / Science Translational Medicine).
A leading commercial provider of liquid biopsy tests designed to provide physicians with clinically actionable information to improve the outcomes of cancer patients announces the commercial availability of its assay for mutations of the NRAS oncogene. The assay can be used to detect and monitor an actionable biomarker associated with multiple cancer types such as metastatic melanoma, colorectal and lung cancer.
The new Target Selector NRAS mutation test is provided by Biocept, Inc (San Diego, CA, USA) and uses the Company's proprietary switch blocker technology, which enriches for oncogene mutations of interest and results in highly sensitive biomarker detection. The Company's liquid biopsy tests are performed in its CLIA-certified, CAP-accredited laboratory. Biocept now offers 15 CLIA-certified liquid biopsy tests utilizing its Target Selector platform to determine the status of key cancer biomarkers listed in the National Comprehensive Cancer Network (NCCN) Guidelines (Fort Washington, PA, USA).
Lyle Arnold, PhD, Biocept's Chief Scientific Officer, said, “Our newest assay combines our proprietary switch blocker technology for improved mutation detection with the power of next generation sequencing, resulting in ultra-high sensitive performance of our liquid biopsy NRAS test. This is our first commercially available assay that utilizes more than one switch blocker in the assay to interrogate multiple genes simultaneously, demonstrating our ability to develop multiplex panels that can analyze several different mutated gene regions in a single test.”
Related Links:
Biocept
National Comprehensive Cancer Network