Real-Time PCR Assay Diagnoses Cutaneous Leishmaniasis
By LabMedica International staff writers
Posted on 14 Jun 2011
A real-time polymerase chain reaction (PCR) assay provides consistent results for cutaneous leishmaniasis within hours of sample receipt.Posted on 14 Jun 2011
The diagnostic test, which is called the SMART Leish Real-Time PCR assay, gives results even when the numbers of parasites in the skin are so low that microscopy and culture results will be negative at day 30.
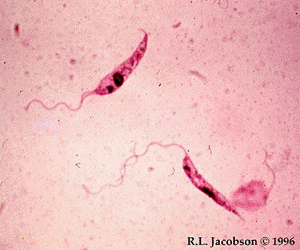
Image: Leishmania promastigotes (Photo courtesy of Dr. R. L. Jacobson, Department of Microbiology and Molecular Genetics, Hebrew University, Jerusalem, Israel).
The SMART Leish PCR assay was developed in partnership among Walter Reed Army Institute of Research (WRAIR; Silver Spring, MD, USA), the Army Medical Research and Materiel Command (Fort Detrick, MD, USA), and the commercial partner, Cepheid USA, Inc. (Sunnyvale, CA, USA) to address the global health issue of cutaneous leishmaniasis that occurred in thousands of US troops serving in Iraq and Afghanistan. The SMART Leish Real-Time PCR assay has now been approved by the US Food and Drug Administration (FDA; Silver Spring, MD, USA).
"In the near future, this assay may be utilized at additional strategically located [Department of Defense] DoD medical facilities, enhancing our capability to provide rapid diagnosis to service members and improve patient management," said Col. Max Grogl, Walter Reed Army Institute of Research's director.
Endemic throughout Africa, Asia, Europe, as well as the Americas, leishmaniasis is a protozoan parasitic disease caused by single-cell organisms transmitted through the bite of a sand fly. Cutaneous leishmaniasis is the most common form of the disease. Though not considered life threatening, the disease can cause disfiguring skin ulcers that take months to heal and cause scarring.
Traditional testing methods require a small tissue sample of the skin ulcer lesion. The samples are examined under microscope and culture to determine the presence of the intracellular form of the Leishmania parasites called amastigotes. These traditional testing methods can require anywhere from 30 minutes to four weeks to produce results.
Related Links:
[US] Army Medical Research and Materiel Command
Walter Reed Army Institute of Research
Cepheid USA














