Gene Mutation Testing Is Critical for Cancer Prognosis
By LabMedica International staff writers
Posted on 22 Sep 2010
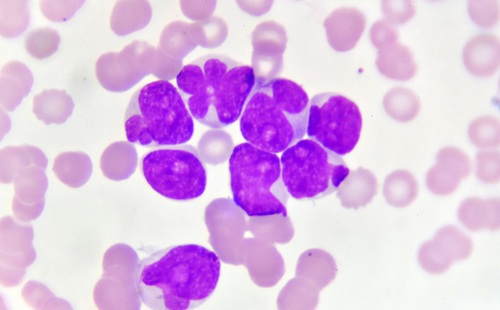
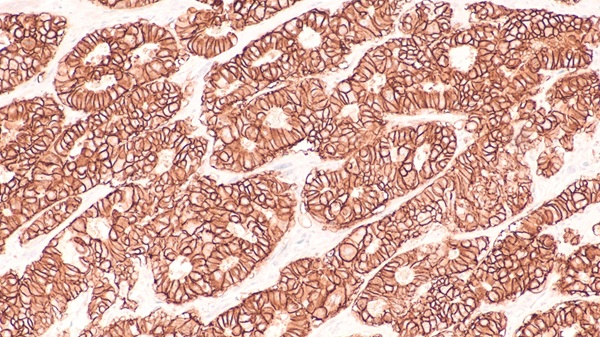
Histological specimens of patients with colon-rectal cancer (CRC) can be tested for gene mutations that will enable physicians to determine the treatment and prognosis. Posted on 22 Sep 2010
Formalin-fixed paraffin-embedded (FFPE) tissue, either on a slide or in a block can be tested for the presence of the Kirsten rat sarcoma viral oncogene (KRAS) or the protooncogene BRAF gene mutations. DNA is extracted from the specimen and mutation specific multiplex polymerase chain reaction (PCR) amplification is performed.
The test requires a minimum of a single 10 µm unstained FFPE slide with a tissue surface area equal or greater than 1 cm2, containing at least 40% abnormal area content. A hematoxylin and eosin (H&E) stained slide should accompany the specimen for pathology assessment. The tests are available in various configurations based on the patient's need or the clinicians' objective. The KRAS 7 mutation testing service allows for detection of the seven clinically relevant KRAS mutations in codons 12 and 13 and the KRAS 12 mutation test configuration expands the KRAS 7 test to detect an additional five KRAS mutations in codon 13.
The Gene Mutation testing service is offered by Asuragen Inc., (Austin, TX, USA), in their College of American Pathologists (CAP)-accredited laboratory. RAS genes are the most common targets for somatic gain-of-function mutations in human cancers. Activating RAS mutations occur in approximately 30% of human cancers and specific RAS genes are mutated in different cancers, including CRC, non-small-cell lung cancer, pancreatic cancer, and other neoplasms. Some mutations in the KRAS gene, in about 40% of colorectal cancer patients, are associated with poor prognosis and lack of response to antiepidermal growth factor receptor therapy. The determination of KRAS mutation status in these tumors is critical when evaluating a patient for such therapy. BRAF is a downstream molecule from KRAS in a signaling pathway involved in cell cycling, and like KRAS, mutations in BRAF are observed in CRC. Mutations in BRAF have been identified in 3% to 12% of CRC patients and are also indicative of poor prognosis. They should be tested for when KRAS testing indicates KRAS wild type.
Carol E. Berry, M.B.A., a vice president of the company, said, "Adding KRAS and BRAF mutational testing to our Clinical Laboratory Improvement Amendments (CLIA) certified laboratory menu further expands our mission in bringing personalized medicine solutions to our partners and patients".
Related Links:
Asuragen Inc.




.jpg)