Rapid One-Step Assay for Field-Based Detection of Asymptomatic Malaria
By LabMedica International staff writers
Posted on 29 Sep 2020
A novel CRISPR-based ultrasensitive assay system was able to detect the four major types of malaria-causing parasites and has been streamlined for use in locations lacking the advanced laboratory equipment and highly trained technicians required for tests such as RT-qPCR, which is used extensively for monitoring the COVID-19 pandemic.Posted on 29 Sep 2020
Asymptomatic carriers of malaria caused by parasites of the Plasmodium species (P. falciparum, P. vivax, P. ovale, and P. malariae) hamper malaria control and eradication. Achieving malaria eradication requires ultrasensitive diagnostics for low parasite density infections (fewer than 100 parasites per microliter blood) that can be used in resource-limited settings (RLS). Molecular methods, such as PCR, have high sensitivity and specificity, but remain high-complexity technologies impractical for RLS.
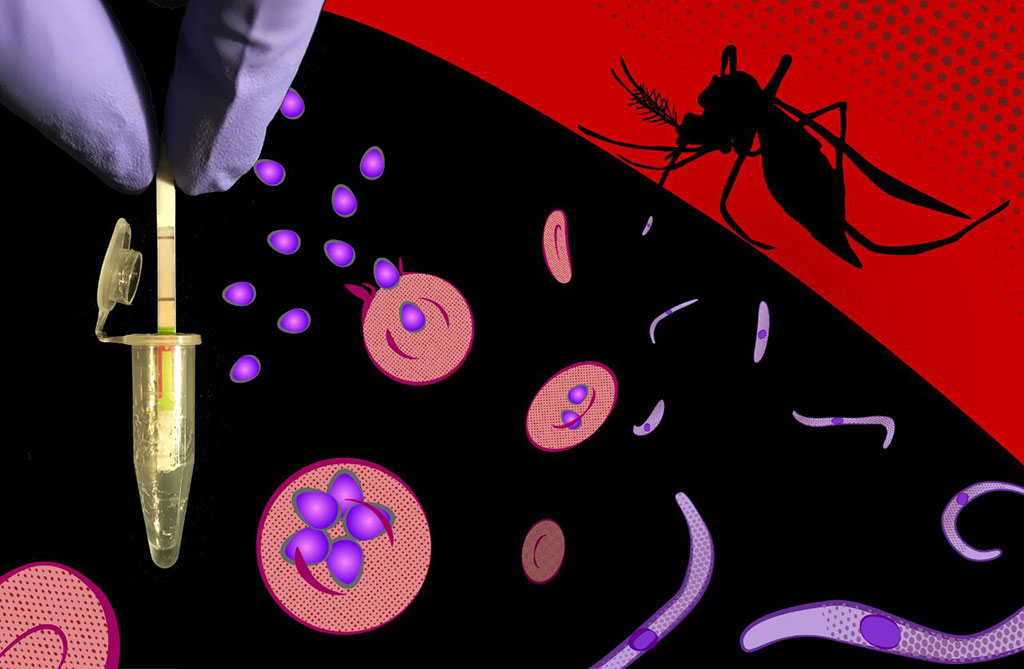
Image: A field-applicable, ultrasensitive diagnostic assay specifically detects DNA and RNA sequences from all Plasmodium species in symptomatic and asymptomatic malaria, and delivers its results fast in simple reporter devices (Photo courtesy of Peter Nguyen, Harvard University)
Investigators at Harvard University (Cambridge, MA, USA) and their collaborators at the Massachusetts Institute of Technology (Cambridge, MA, USA) and the Wyss Institute for Biologically Inspired Engineering (Cambridge, MA) reported the development of a CRISPR-based diagnostic tool for ultrasensitive detection and differentiation of the four Plasmodium parasites, using the nucleic acid detection platform SHERLOCK (specific high-sensitivity enzymatic reporter unlocking).
The investigators modified the SHERLOCK protocol to incorporate the CRISPR-Cas12a enzyme, which they programmed to become activated by a guide RNA that bound to a specific target nucleic acid target sequence, in this case a sequence from one of the four Plasmodium species. Activated Cas12a then non-specifically cleaved any single-stranded DNA strand in its vicinity with an extremely high turn-over rate of about 1,250 collateral cleavage reactions per second.
The complete assay platform was expanded to comprise a 10-minute SHERLOCK parasite rapid extraction protocol, followed by the SHERLOCK diagnostic protocol for 60 minutes, which enabled Plasmodium species-specific detection via fluorescent or lateral flow strip readout. The assay was compatible with different sample types, such as whole blood, plasma, serum, and dried blood; and all components required for amplification, Cas12a activation, and signal generation were lyophilized in a single test tube that functioned as a "one-pot-reaction" following reconstitution of the reagents when mixed with a patient sample.
Performance of the simplified field-ready SHERLOCK diagnostic was evaluated using simulated whole blood, serum, and dried blood spot (DBS) samples, as well as clinical samples from patients with P. falciparum and P. vivax infections. Results revealed that the assay was capable of detecting fewer than two parasites per microliter blood, a limit of detection suggested by the World Health Organization (WHO). The P. falciparum and P. vivax assays exhibited 100% sensitivity and specificity on clinical samples (five P. falciparum and 10 P. vivax samples).
"This field-ready SHERLOCK diagnostic malaria assay surpasses the sensitivity and specificity requirements set by the WHO for a desired test that can be used to detect low parasite density in asymptomatic carriers of all major Plasmodium species," said senior author Dr. James Collins, professor of medical engineering and science at the Massachusetts Institute of Technology. "Its highly streamlined design could provide a viable solution to the present diagnostic bottleneck on the path to eliminate malaria, and more generally enabling malaria surveillance in low-resource settings."
The rapid SHERLOCK assay for malaria parasites was described in the September 21, 2020, online edition of the journal Proceedings of the National Academy of Sciences of the United States of America.
Related Links:
Harvard University
Massachusetts Institute of Technology
Wyss Institute for Biologically Inspired Engineering














