Newborn Cytomegalovirus Test Cleared for Marketing
By LabMedica International staff writers
Posted on 20 Dec 2018
Cytomegalovirus or CMV is a common virus that infects people of all ages. In the USA, nearly one in three children are already infected with CMV by age five and over half of adults by age 40 have been infected with CMV.Posted on 20 Dec 2018
Most people infected with CMV show no signs or symptoms. That is because a healthy person’s immune system usually keeps the virus from causing illness. However, CMV infection can cause serious health problems for people with weakened immune systems and for unborn babies. Only about one in five babies with congenital CMV infection will be sick from the virus or have long-term health problems.
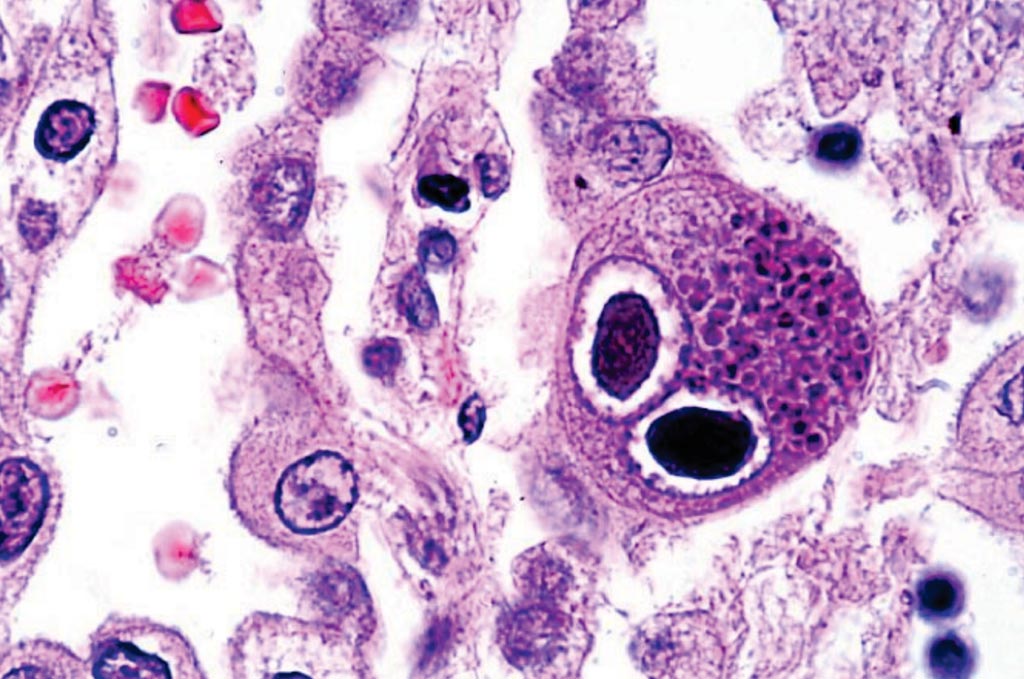
Image: A histopathology of cytomegalovirus infection in the lung showing typical owl-eye inclusions; FDA clears new CMV test for newborns (Photo courtesy of Danny Wiedbrauk, PhD).
The US Food and Drug Administration (FDA, Silver Springs, MD, USA) announced that it has completed the de novo premarket review and cleared for marketing Meridian Bioscience's (Cincinnati, OH, USA) cytomegalovirus assay for newborns called the Alethia CMV Assay Test System. A prospective clinical study showed the Alethia test correctly identified 1,472 out of 1,475 CMV negative samples. There were three false positive tests, and five saliva specimens correctly identified as positive. Meanwhile 34 archived positive specimens were also correctly identified. The Alethia CMV test system enables detection of CMV DNA from saliva swabs of newborn babies who are less than 21 days old. The FDA specified that test results must be used in conjunction with the results of other diagnostic tests and clinical information.
Timothy Stenzel, MD, PhD, the director of the Office of In Vitro Diagnostics and Radiological Health in the FDA’s Center for Devices and Radiological Health said, “Although most people who become infected with cytomegalovirus face little to no risk of serious illness, the virus has the potential to cause serious illness for people with weak immune systems and in newborn babies.”




.jpg)









