Rapid Syphilis Test Evaluated as Tool for Public Health Programs
By LabMedica International staff writers
Posted on 09 Nov 2016
A rapid commercial treponemal syphilis-screening test is a new tool for public health programs to combat increasing syphilis rates, specifically among persons without a prior syphilis infection.Posted on 09 Nov 2016
The Syphilis Health Check (SHC) test can be performed by non-laboratorian healthcare personnel and results are available in 10 minutes and have recently received a Clinical Laboratory Improvement Amendments waiver.
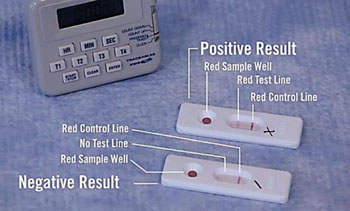
Image: The Syphilis Health Check test is a qualitative rapid membrane immune-chromatographic assay for the detection of Treponema pallidum (syphilis) antibodies in human whole blood, serum and plasma (Photo courtesy of Trinity Biotech).
Scientists at the Florida Department of Health (Tampa, FL, USA) in collaboration with The Centers of Disease Control and Prevention (CDC, Atlanta, GA, USA) evaluated the performance of SHC in comparison with treponemal and nontreponemal tests routinely used in its sexually transmitted disease (STD) clinic. For this evaluation, patients seeking sexually transmitted diseases (STD) testing at the Florida Department of Health STD clinic in Escambia County during March 11 to April 21, 2016, were tested for syphilis using the SHC on blood specimens obtained by fingerstick; a venous blood specimen was drawn concurrently and submitted for treponemal and testing at the state public health laboratory.
Trep-Sure qualitative enzyme immunoassay (EIA) reference treponemal test was used as the standard for “true” positive or negative treponemal test results, when evaluating the SHC. The rapid plasma reagin (RPR) card for syphilis was also tested. The SHC was used to screen 202 patients for syphilis. Among these patients, 171 (85%) were nonreactive on all syphilis tests (SHC, EIA, and RPR), 26 (13%) had a reactive SHC, and five (2%) had a nonreactive SHC, but had one or more reactive tests at the state laboratory.
Among the 26 reactive SHCs, 10 (38%) had a reactive EIA (six had a reactive RPR), and 16 (62%) were not confirmed by EIA or RPR at the state laboratory. For the six reactive SHC patients with reactive EIA and reactive RPR, three were staged as secondary syphilis, one as primary syphilis, one as early latent syphilis, and one was a previously treated positive with no increase in titer since last testing. Among the five specimens that were reactive on other tests but SHC nonreactive, only one was both RPR (1:8 serum dilution) and EIA reactive. The sensitivity of SHC was 71.4% when compared with the Trep-Sure (EIA) reference treponemal test. The specificity of the SHC compared with the reference treponemal test was 91.5%. The study was published on October 28, 2016, in the journal Morbidity and Mortality Weekly Report.
Related Links:
Florida Department of Health
The Centers of Disease Control and Prevention




.jpg)









