Immunochromatographic Test Developed for Visceral Leishmaniasis
By LabMedica International staff writers
Posted on 16 Jul 2015
The available diagnostic tests for visceral leishmaniasis are either invasive and require considerable expertise such as the parasitological demonstration of the parasite in tissue smears or unable to distinguish between past and active infection as in serological methods.Posted on 16 Jul 2015
A lateral flow assay in the form of an immunochromatographic test (ICT) device based on the detection of a circulating Leishmania antigen using monoclonal antibodies (mAbs) has been developed that will allow for early case detection followed by adequate treatment is essential to the control of VL.
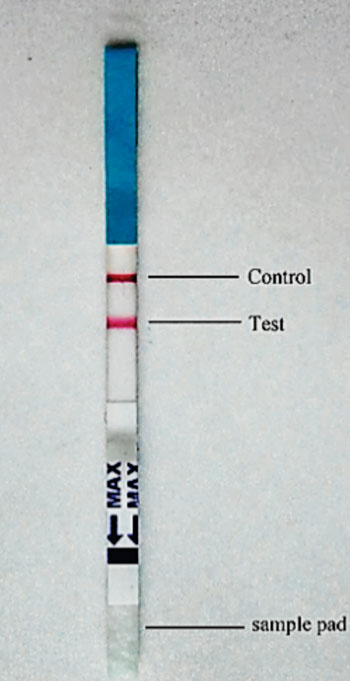
Image: An immunochromatographic strip tested with a serum sample from a visceral leishmaniasis patient, both the control and the test line turned pink (Photo courtesy of Chinese National Institute of Parasitic Diseases).
Scientists at the Chinese National Institute of Parasitic Diseases (Shanghai, China) produced mAbs that were used to develop an ICT as a sandwich assay for the detection of circulating antigen in serum and blood samples. The ICT was evaluated with serum samples from VL patients living in VL endemic areas in China, and with 156 serum samples from patients with other diseases as well as 78 serum samples from healthy donors. The commercially available Kalazar Detect Rapid Test for visceral leishmaniasis (InBios International, Inc.; Seattle, USA) was used as a comparison assay. This diagnostic tool is an ICT for the detection of anti-Leishmania donovani antibodies (anti-k39 antibody) in human serum.
The newly developed ICT was evaluated with 213 serum samples from VL patients. Of these, 95.8% (204) serum samples gave a positive reaction. With respect to the different endemic VL areas, serum samples showed a positive reaction from anthroponotic type of VL (AVL) in 63/64 (98.4%), from desert sub-type of zoonotic VL (DST-ZVL) in 66/71 (93.0%), and in 75/78 (96.2%) from mountain sub-type of zoonotic VL (MST-ZVL) endemic regions. All VL patients were diagnosed by microscopic examinations of bone marrow smears. The newly developed ICT detecting a circulating leishmanial antigen performed as well as the commercially available Kalazar Detect Rapid Test detecting an anti-leishmanial antibody.
The authors concluded that the newly developed ICT is an easy to use and more accurate diagnostic tool that fulfils the performance and operational characteristics required for VL case detection under field and laboratory conditions. As the ICT detects a circulating antigen, it will also be useful in monitoring treatment success and diagnosing VL in immunocompromised patients. The study was published on June 30, 2015, in the journal Public Library of Science Neglected Tropical Diseases.
Related Links:
Chinese National Institute of Parasitic Diseases














