Oral HIV Test Could Reduce Infection Rates
By LabMedica International staff writers
Posted on 25 Mar 2015
The performance and assessment of an oral test has been compared with standard serological testing of an enzyme immunoassay (EIA) in the clinical setting, and its acceptability to individuals at high risk of human immunodeficiency virus (HIV) infection. Posted on 25 Mar 2015
Barriers to being tested may be overcome by the use of HIV rapid tests that are reliable (sensitive and specific), efficient (render results quickly) and acceptable to the individuals being tested. However, it is important to remember that rapid tests are screening tests and do not replace diagnostic serological tests.
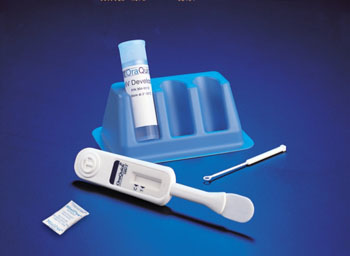
Image: The OraQuick Advance Rapid HIV-1/2 Antibody Test (Photo courtesy of Orasure Technologies).
Scientists at Albion Centre, South Eastern Sydney Local Health District (Sydney, NSW, Australia) performed a cross-sectional study of 1,074 men who have sex with men (MSM) and individuals aged 18 years or older at high risk of acquiring HIV infection who attended five public HIV or sexual health services, two general practices and one community clinic in Sydney from 1 January to December 31, 2013.
The testing was done with a single OraQuick Advance Rapid HIV-1/2 Antibody Test (ORT; Orasure Technologies, Bethlehem, PA, USA) plus a confirmatory fourth-generation EIA, either the Elecsys HIV Combi PT (Roche Diagnostics, Basel, Switzerland) or Architect HIV Ag/Ab Combo (Abbott Diagnostics; Princeton, NJ, USA) assay. The OraSure test does not use saliva but uses a fluid called oral mucosal transudate, which lives in the cheeks and gums The ORT two-step process involved sweeping the test swab across participants' upper and lower gum lines and submerging it in a developer solution vial and this yielded an HIV-1/2 antibody result in a minimum of 20 minutes.
The results confirmed that the ORT is both sensitive and specific when compared with the standard fourth generation HIV enzyme immunoassay (EIA), and also highly acceptable to the patients. Three-quarters (74.0%) of the participants found the ORT less stressful than giving a conventional blood sample. Nearly all (99.2%) said they would have an ORT again and 99.4% said they would recommend ORT to their peers. However, they were not asked whether having to pay for the test would affect their decision to be tested again.
The authors concluded that HIV rapid tests such as the ORT can play a key role in early diagnosis, initiation of antiretroviral therapy and preventing transmission, particularly among MSM and individuals at high risk. This will deliver clear individual and public health benefits. As is the case in the USA, they believe the ORT is an appropriate alternative rapid test to tackle Australia's HIV epidemic. The study was published on March 9, 2015, in the Medical Journal of Australia.
Related Links:
South Eastern Sydney Local Health District
Orasure Technologies
Roche Diagnostics














