Digital Aging Twin Quantifies Biological Aging Across Multiple Organ Systems
Posted on 13 May 2026
Chronological age often fails to capture the wide variability in physiological decline among adults, limiting risk stratification and long-term monitoring. Clinical laboratories also lack standardized tools that can generate reproducible measures of aging across multiple organ systems. Because these systems may age at different rates, objective and interpretable assessment has remained difficult. A new study shows that a computational Digital Aging Twin can quantify both individual and organ-specific aging across adulthood.
Researchers at the Chinese Academy of Sciences’ Institute of Zoology and the China National Center for Bioinformation (Chinese Academy of Sciences), together with Xuanwu Hospital of Capital Medical University and seven partner institutions, developed the Digital Aging Twin to estimate biological age and track organ-level aging rates. The framework advances aging assessment from descriptive markers to quantitative, interpretable modeling. It integrates clinical, imaging, functional, and molecular readouts to generate multiscale aging “clocks.”
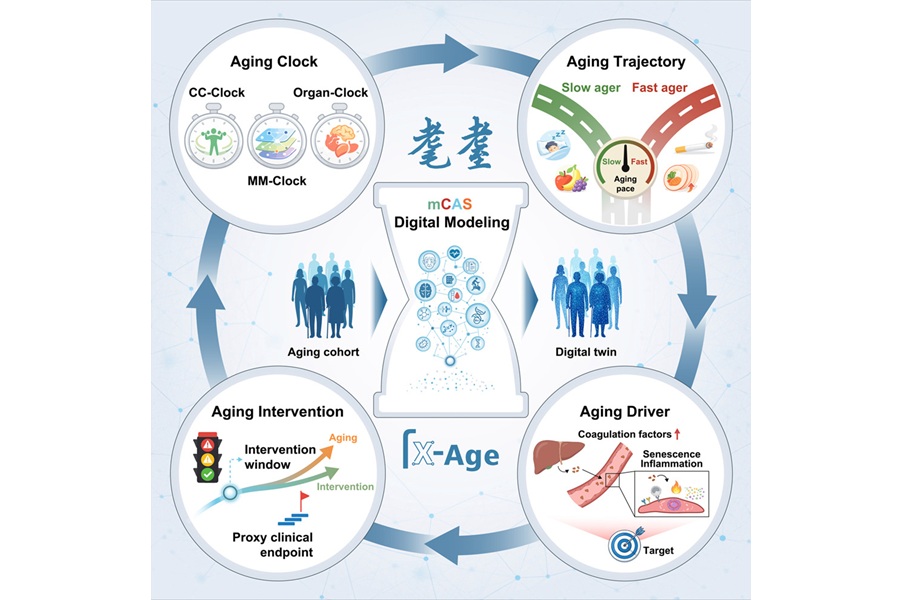
The system is organized into three tiers. The first is a core capacity clock that combines 240 physiological indicators to reflect overall functional decline. The second is a multimodal clock that uses deep learning to integrate multiple molecular data layers, including DNA methylation, RNA transcripts, proteins, metabolites, and gut microbiome profiles, while weighting the most informative inputs. The third consists of organ-specific clocks for the brain, liver, lungs, muscles, blood vessels, and skin, built from clinical markers, plasma proteins, and imaging features.
Study data were drawn from mCAS (multicentric Chinese Aging Standardized), a standardized multicenter cohort of 2,019 healthy individuals aged 18 to 91 from Beijing, Quzhou, Ningbo, and Nanchang. More than one billion high-quality data points were collected across clinical tests, cognitive and motor assessments, brain and retinal imaging, gait analysis, and several layers of molecular data, including DNA methylation, RNA transcripts, proteins, metabolites, and gut microbiomes. While the current framework is based on cross-sectional data, the program is being refined with longitudinal follow-up.
The multimodal clock predicted chronological age with a mean absolute error of just 3.87 years, outperforming single-omics clocks. Notably, the analyses revealed that organs age asynchronously, with liver aging showing an inflection near age 40 and brain aging accelerating around age 50. Broader nonlinear waves of change were also observed between ages 40–50 and 60–70.
Additional experiments using plasma proteomics, histology, cell culture, and animal models identified liver-derived coagulation factors, particularly F13B, along with F9 and F10, as direct drivers of vascular and systemic aging. In mice, F13B accelerated aging across multiple tissues, while these factors also induced senescence-related changes in human aortic endothelial cells.
For clinical practicality, the team built “proxy clocks” using 100–108 plasma proteins that closely matched the comprehensive clocks. Lifestyle associations were also noted: greater fruit intake, consistent sleep routines, and moderate walking correlated with slower aging, while smoking, insufficient sleep, and high meal frequency correlated with faster aging.
The findings, published in Cell on May 8, 2026, represent the first proof-of-concept milestone of China’s X-Age Project and provide a standardized, quantifiable, and interpretable system to localize and compare aging rates across organ systems.