Epigenetic Signals and Blood Markers Aid Chronic Fatigue Syndrome Diagnosis
Posted on 27 Apr 2026
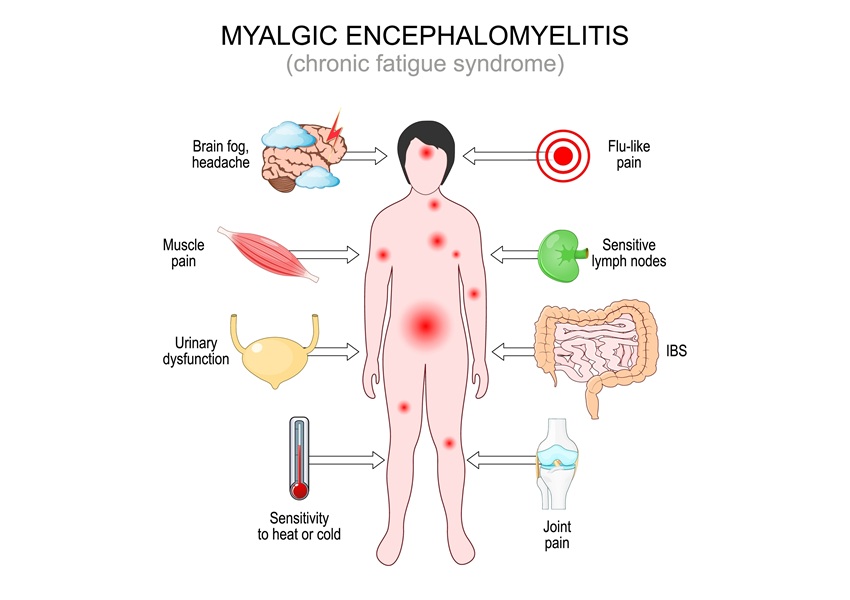
Myalgic encephalomyelitis (ME), also known as chronic fatigue syndrome, presents with heterogeneous symptoms and a hallmark of post‑exertional malaise that complicate clinical assessment. Many patients experience multisystem involvement with fluctuating severity, and overlap with conditions such as fibromyalgia can blur diagnostic boundaries. Clinically useful biomarkers remain limited, hindering accurate classification and targeted study designs. Researchers now describe epigenetic signals and circulating biomarkers that may sharpen differentiation and track disease severity.
At Université de Montréal, investigators highlight molecular approaches centered on epigenetics, circulating microRNA patterns, and the blood protein SMPDL3B to improve identification of myalgic encephalomyelitis. Specific circulating microRNA signatures are reported to accurately distinguish the condition from fibromyalgia. SMPDL3B concentration in blood is directly linked to symptom intensity, positioning it as a candidate severity marker.
The group emphasizes epigenetics—environmentally driven changes in gene expression without altering the DNA sequence—as a promising framework for understanding disease mechanisms. A recent study published in the journal DNA points to factors such as infections and toxins as potential reprogrammers of key biological pathways. In about 75% of cases, ME onset follows a viral illness, with triggers that include Epstein–Barr virus, influenza, West Nile virus, chikungunya, Zika, and SARS‑CoV‑2.
Building on the SMPDL3B signal, researchers previously identified the enzyme responsible for elevated circulating levels in a study published last year. They are exploring inhibition of this enzyme, noting that some diabetes medications could be repurposed alongside myo‑inositol supplementation. Clinical trials are being planned to evaluate this therapeutic strategy.
The work originates from Université de Montréal and the CHU Sainte‑Justine Research Center. The authors underscore that measurable biological abnormalities, particularly neuroinflammation in many patients, validate the condition’s pathobiology. They call for targeted investigations that combine advanced molecular analyses with dynamic tests capable of reproducing post‑exertional malaise, especially in the most severely affected individuals who are often excluded from conventional research protocols.
“ME is not a single disease but rather a spectrum comprising several subgroups. Accurate diagnosis is difficult because ME and fibromyalgia present with very similar symptoms that can fluctuate over time. Our molecular analyses have shown that up to 40% of individuals diagnosed with ME are in fact suffering from fibromyalgia—and it's possible to have both conditions at the same time,” said Alain Moreau, professor in the Department of Biochemistry and Molecular Medicine and the Department of Stomatology at Université de Montréal and researcher at the CHU Sainte‑Justine Research Center.
“ME remains difficult to define, but recent advances are gradually changing the landscape. With the rise of long COVID, this long‑marginalized condition is now a scientific priority—a shift that could finally transform patient care,” said Moreau.
Related Links
CHU Sainte‑Justine Research Center
Université de Montréal