Microenvironment Biomarkers Could Enable Early Lung Cancer Detection
Posted on 25 Apr 2026
Early detection of lung cancer remains difficult because the first cellular changes unfold within a shifting local niche. Clarifying how mutant cells reshape surrounding tissue could aid diagnosis at stages when disease is more treatable. Evidence in model systems indicates some of these earliest shifts can be reversed if intercepted. A new study shows how oncogene-bearing lung cells rapidly reprogram fibroblasts and immune components to establish a tumor-permissive microenvironment.
Memorial Sloan Kettering Cancer Center (MSK) investigators and colleagues mapped the earliest steps of tumor initiation in lung tissue and the signals that remodel the “tumor microenvironment.” The team combined mouse models carrying KRAS mutations with three-dimensional (3D) lung assembloids generated from mouse and human tissue. Using experiments that tracked individual tumor cells from the moment they acquired mutations, the researchers delineated a multi-cellular cascade that enables nascent lesions to progress.
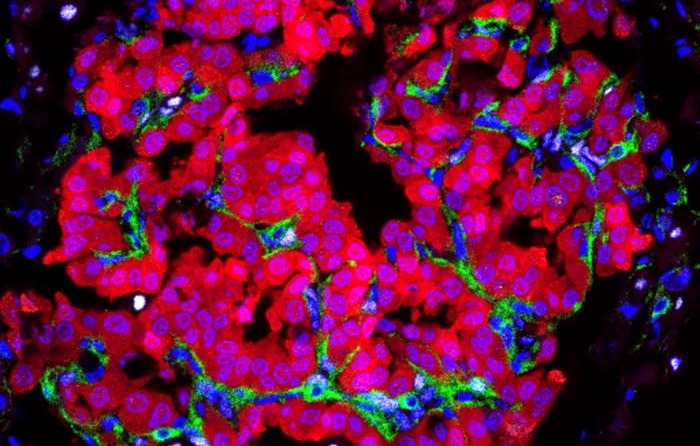
The work identifies a three-step communication loop. First, KRAS-mutant epithelial cells adopt a regenerative state and secrete high levels of amphiregulin (AREG), a distress signal sensed by nearby healthy cells. Second, fibroblasts interpret this signal as injury, become fibrotic, and lay down a supporting scaffold similar to wound-healing responses.
In the third step, these activated fibroblasts promote tumor growth and reshape the immune microenvironment; macrophages expand locally but suppress effective antitumor responses, while cells such as neutrophils and regulatory T cells are recruited, favoring immune dampening rather than tumor clearance. Together, these interactions create a self-reinforcing loop in which the remodeled environment sustains the mutant cells’ regenerative program and supports continued tumor growth.
Disrupting this dialogue abrogated tumor development in models. Using an epidermal growth factor receptor (EGFR) inhibitor already approved for advanced lung cancer, the team blocked AREG-mediated signaling, keeping fibroblasts and immune cells in normal states and markedly impairing tumor formation. Similar effects were observed when the AREG gene was deleted from mutant cells, preventing activation of the surrounding stromal and immune compartments.
In early lesions, inhibition of KRAS activity reversed many of the microenvironmental alterations, indicating that these early changes remain plastic.
To assess clinical relevance, the researchers analyzed tissue samples from patients with early-stage lung adenocarcinoma and observed the same spatial arrangement seen in models, with AREG-high tumor cells adjacent to fibrotic fibroblasts. Because such samples provide only a single timepoint, the team developed patient-derived lung stem cell assembloids to model tumor initiation dynamically.
By introducing KRAS mutations into these three-dimensional cultures, they were able to trace early tumor development in real time and confirm induction of the same AREG-driven regenerative program. Co-culture experiments with normal human lung fibroblasts reproduced fibroblast activation, which could again be blocked with EGFR inhibition. Similar patterns were observed when modeling EGFR mutations, indicating the mechanism is not limited to KRAS-driven disease.
The authors note that comparable microenvironmental remodeling has been reported in esophageal and pancreatic cancers, supporting a broader paradigm in which early tumor cells rapidly construct a supportive niche. These findings point to potential biomarkers for earlier detection and may be particularly relevant for high-risk populations, including long-term smokers or individuals with genetic predispositions who may harbor precancerous lesions.
In addition, the results may help explain clinical observations in which patients with AREG-high tumors respond to EGFR inhibitors despite lacking EGFR mutations, suggesting new opportunities for earlier identification and intervention. The study was published April 22 in Nature.
“The key message we’d really like to deliver is: When tumors first emerge, they hijack the regenerative program, and they interact with healthy cells to generate a microenvironment supporting the cancer. This determines whether these tumor cells sustain and progress and it happens at a very early stage,” said Joo-Hyeon Lee, Ph.D.,
"Cancer cells don't exist in isolation," says Scott Lowe, Ph.D., scientific director of the Ecosystems Project. "From the very beginning, they're engaging in complex interactions with their neighbors and with the immune system. This study is a great example of the type of fundamental scientific discovery that will aid the development of innovative new treatments that could benefit large numbers of cancer patients."
Related Links
Memorial Sloan Kettering Cancer Center














