Label-Free Microfluidic Device Enriches Tumor Cells and Clusters from Pleural Effusions
Posted on 25 Apr 2026
Diagnosing malignancy from pleural effusion remains challenging because tumor cells are rare and clusters are easily disrupted during processing. Conventional cytology can miss malignant tumor cells and often yields limited insight into metastatic potential. Label-dependent enrichment can damage cells and obscure surface markers, while many label-free methods struggle with throughput and purity. A new study shows a cascaded microfluidic approach that enriches both single malignant tumor cells and intact clusters from pleural effusions in a fast, label-free workflow.
Researchers at Southeast University (Nanjing, China), working with Wuxi University of Technology and Zhongda Hospital of Southeast University, developed a cascaded inertial microfluidic device to recover single malignant tumor cells (MTCs) and intact malignant tumor cell clusters (MTCCs) from pleural effusions of lung cancer patients. The platform is designed to make malignancy assessment more informative by preserving cluster architecture while sharply reducing background blood cells. The method processes large volumes rapidly without labeling.
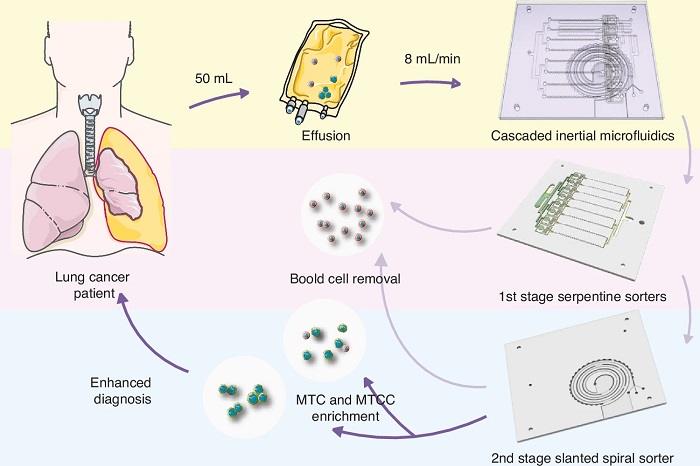
The workflow links two size-based sorting stages. Parallelized serpentine channels first deplete most background blood cells at high throughput by driving smaller cells toward sidewalls for removal while retaining larger targets in the core stream. A second slanted spiral channel then separates single tumor cells from larger clusters through size-dependent inertial focusing governed by the balance among inertial lift, Dean drag, and locally induced vortices.
In model particle experiments, the device recovered 91.8% ± 6.6% of 25 μm particles representing clusters and 87.4% ± 7.4% of 15 μm particles representing single cells. In simulated samples containing A549 lung cancer cells and white blood cells (WBCs), overall A549 recovery reached 81.7% ± 1.2% with 76.1% ± 1.3% purity; single-cell fractions collected at outlet 3 achieved 75.9% ± 1.3% recovery and 75.8% ± 1.6% purity, while cluster-rich fractions at outlet 4 reached 79.4% ± 3.4% purity. In three patient pleural effusion samples, the chip processed 50 mL in 6.5 minutes at 8 mL/min, yielding 68% purity for single MTCs and 35% purity for intact MTCCs. Tumor cells were identified by immunofluorescence as DAPI+/Pan-CK+/CD45−.
The study was published in Microsystems & Nanoengineering on March 23, 2026. The authors describe the chip as label-free, requiring no external fields or complex instrumentation, and compatible with standard staining and microscopic analysis. They also note that its multilayer polymer construction is suited to scalable, relatively low-cost manufacturing.
Clinical validation remains preliminary, limited to three patient samples, and larger cohorts are needed to establish diagnostic sensitivity and prognostic value. The approach is presented as a route toward faster cytologic support, improved recovery of fragile tumor clusters, and more actionable malignancy assessment in pleural effusions.